Articles
- Page Path
- HOME > Epidemiol Health > Volume 45; 2023 > Article
-
COVID-19
Original Article
Korea Seroprevalence Study of Monitoring of SARS-COV-2 Antibody Retention and Transmission (K-SEROSMART): findings from national representative sample -
Jina Han1
 , Hye Jin Baek1
, Hye Jin Baek1 , Eunbi Noh2
, Eunbi Noh2 , Kyuhyun Yoon3
, Kyuhyun Yoon3 , Jung Ae Kim4
, Jung Ae Kim4 , Sukhyun Ryu5
, Sukhyun Ryu5 , Kay O Lee6
, Kay O Lee6 , No Yai Park7
, No Yai Park7 , Eunok Jung8
, Eunok Jung8 , Sangil Kim9
, Sangil Kim9 , Hyukmin Lee10
, Hyukmin Lee10 , Yoo-Sung Hwang11
, Yoo-Sung Hwang11 , Jaehun Jung12
, Jaehun Jung12 , Hun Jae Lee13
, Hun Jae Lee13 , Sung-il Cho3,14
, Sung-il Cho3,14 , Sangcheol Oh15
, Sangcheol Oh15 , Migyeong Kim16
, Migyeong Kim16 , Chang-Mo Oh17
, Chang-Mo Oh17 , Byengchul Yu18
, Byengchul Yu18 , Young-Seoub Hong19
, Young-Seoub Hong19 , Keonyeop Kim20
, Keonyeop Kim20 , Sunjae Jung21
, Sunjae Jung21 , Mi Ah Han22
, Mi Ah Han22 , Moo-Sik Lee5
, Moo-Sik Lee5 , Jung-Jeung Lee23
, Jung-Jeung Lee23 , Young Hwangbo24
, Young Hwangbo24 , Hyeon Woo Yim25
, Hyeon Woo Yim25 , Yu-Mi Kim26,27
, Yu-Mi Kim26,27 , Joongyub Lee28,29
, Joongyub Lee28,29 , Weon-Young Lee30
, Weon-Young Lee30 , Jae-Hyun Park31
, Jae-Hyun Park31 , Sungsoo Oh32
, Sungsoo Oh32 , Heui Sug Jo33
, Heui Sug Jo33 , Hyeongsu Kim34
, Hyeongsu Kim34 , Gilwon Kang35
, Gilwon Kang35 , Hae-Sung Nam36
, Hae-Sung Nam36 , Ju-Hyung Lee37
, Ju-Hyung Lee37 , Gyung-Jae Oh38
, Gyung-Jae Oh38 , Min-Ho Shin39
, Min-Ho Shin39 , Soyeon Ryu22
, Soyeon Ryu22 , Tae-Yoon Hwang40
, Tae-Yoon Hwang40 , Soon-Woo Park41
, Soon-Woo Park41 , Sang Kyu Kim42
, Sang Kyu Kim42 , Roma Seol43
, Roma Seol43 , Ki-Soo Park44
, Ki-Soo Park44 , Su Young Kim45
, Su Young Kim45 , Jun-wook Kwon46
, Jun-wook Kwon46 , Sung Soon Kim47
, Sung Soon Kim47 , Byoungguk Kim48
, Byoungguk Kim48 , June-Woo Lee48
, June-Woo Lee48 , Eun Young Jang48
, Eun Young Jang48 , Ah-Ra Kim48
, Ah-Ra Kim48 , Jeonghyun Nam48
, Jeonghyun Nam48 , The Korea Community Health Survey Group, Soon Young Lee1
, The Korea Community Health Survey Group, Soon Young Lee1 , Dong-Hyun Kim49
, Dong-Hyun Kim49
-
Epidemiol Health 2023;45:e2023075.
DOI: https://doi.org/10.4178/epih.e2023075
Published online: August 17, 2023
1Department of Preventive Medicine and Public Health, Ajou University School of Medicine, Suwon, Korea
2National Radiation Emergency Medical Center, Korea Institute of Radiological and Medical Sciences, Seoul, Korea
3Institute of Health and Environment, Seoul National University, Seoul, Korea
4Department of Nursing, Kyungmin University, Uijeongbu, Korea
5Department of Preventive Medicine, Konyang University College of Medicine, Daejeon, Korea
6Gallup Korea, Seoul, Korea
7Graduate School of Public Health, Inje University, Seoul, Korea
8Department of Mathematics, Konkuk University, Seoul, Korea
9Department of Internal Medicine, College of Medicine, The Catholic University, Seoul, Korea
10Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea
11Seegene Medical Foundation, Seoul, Korea
12Department of Preventive Medicine, Gachon University College of Medicine, Incheon, Korea
13Department of Social and Preventive Medicine, Inha University College of Medicine, Incheon, Korea
14Department of Public Health Science, Graduate School of Public Health, Seoul National University, Seoul, Korea
15MAPO-gu Public Health Center, Seoul, Korea
16GUNPO-si Public Health Center, Gunpo, Korea
17Department of Preventive Medicine, Kyung Hee University School of Medicine, Seoul, Korea
18Department of Preventive Medicine, Kosin University College of Medicine, Busan, Korea
19Department of Preventive Medicine, Dong-A University College of Medicine, Busan, Korea
20Department of Preventive Medicine, Kyungpook National University School of Medicine, Daegu, Korea
21Department of Preventive Medicine, Yonsei University College of Medicine, Seoul, Korea
22Department of Preventive Medicine, Chosun University College of Medicine, Gwangju, Korea
23Department of Preventive Medicine, Keimyung University School of Medicine, Daegu, Korea
24Department of Preventive Medicine, Soonchunhyang University College of Medicine, Cheonan, Korea
25Department of Preventive Medicine, College of Medicine, The Catholic University, Seoul, Korea
26Department of Preventive Medicine, Hanyang University College of Medicine, Seoul, Korea
27School of Public Health, Hanyang University, Seoul, Korea
28Department of Preventive Medicine, Seoul National University College of Medicine, Seoul, Korea
29Institute of Health Policy and Management, Medical Research Center, Seoul National University, Seoul, Korea
30Department of Preventive Medicine, Chung-Ang University College of Medicine, Seoul, Korea
31Department of Preventive Medicine, Sungkyunkwan University School of Medicine, Suwon, Korea
32Department of Occupational & Environmental Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
33Department of Health Policy and Management, Kangwon National University School of Medicine, Chuncheon, Korea
34Department of Preventive Medicine, Konkuk University School of Medicine, Seoul, Korea
35Department of Health Information and Management, Chungbuk National University College of Medicine, Cheongju, Korea
36Department of Preventive Medicine, Chungnam National University College of Medicine, Daejeon, Korea
37Department of Preventive Medicine, Jeonbuk National University Medical School, Jeonju, Korea
38Department of Preventive Medicine, Wonkwang University School of Medicine, Iksan, Korea
39Department of Preventive Medicine, Chonnam National University Medical School, Hwasun, Korea
40Department of Preventive Medicine & Public Health, Yeungnam University College of Medicine, Gyeongsan, Korea
41Department of Preventive Medicine, Daegu Catholic University School of Medicine, Gyeongsan, Korea
42Department of Preventive Medicine, Dongguk University College of Medicine, Gyeongju, Korea
43Department of Preventive Medicine, Inje University College of Medicine, Busan, Korea
44Department of Preventive Medicine, Institute of Health Sciences, Gyeongsang National University College of Medicine, Jinju, Korea
45Department of Preventive Medicine, Jeju National University School of Medicine, Jeju, Korea
46National Institute of Health, Korea Disease Control and Prevention Agency, Cheongju, Korea
47Center for Vaccine Research, National Institute of Infectious Diseases, Korea Disease Control and Prevention Agency, Cheongju, Korea
48Division of Vaccine Clinical Research, Center for Vaccine Research, National Institute of Infectious Diseases, Cheongju, Korea
49Department of Social and Preventive Medicine, Hallym University College of Medicine, Chuncheon, Korea
- Correspondence: Dong-Hyun Kim Department of Social and Preventive Medicine, Hallym University College of Medicine, 1 Hallymdaehak-gil, Chuncheon 24252, Korea E-mail: dhkims@hallym.ac.kr
- Co-correspondence: Soon Young Lee Department of Preventive Medicine and Public Health, Ajou University School of Medicine, 206 World cup-ro, Yeongtong-gu, Suwon 16499, Korea E-mail: solee5301@gmail.com
- *The Korea Community Health Survey Group researchers are listed in the Acknowledgments section.
© 2023, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES
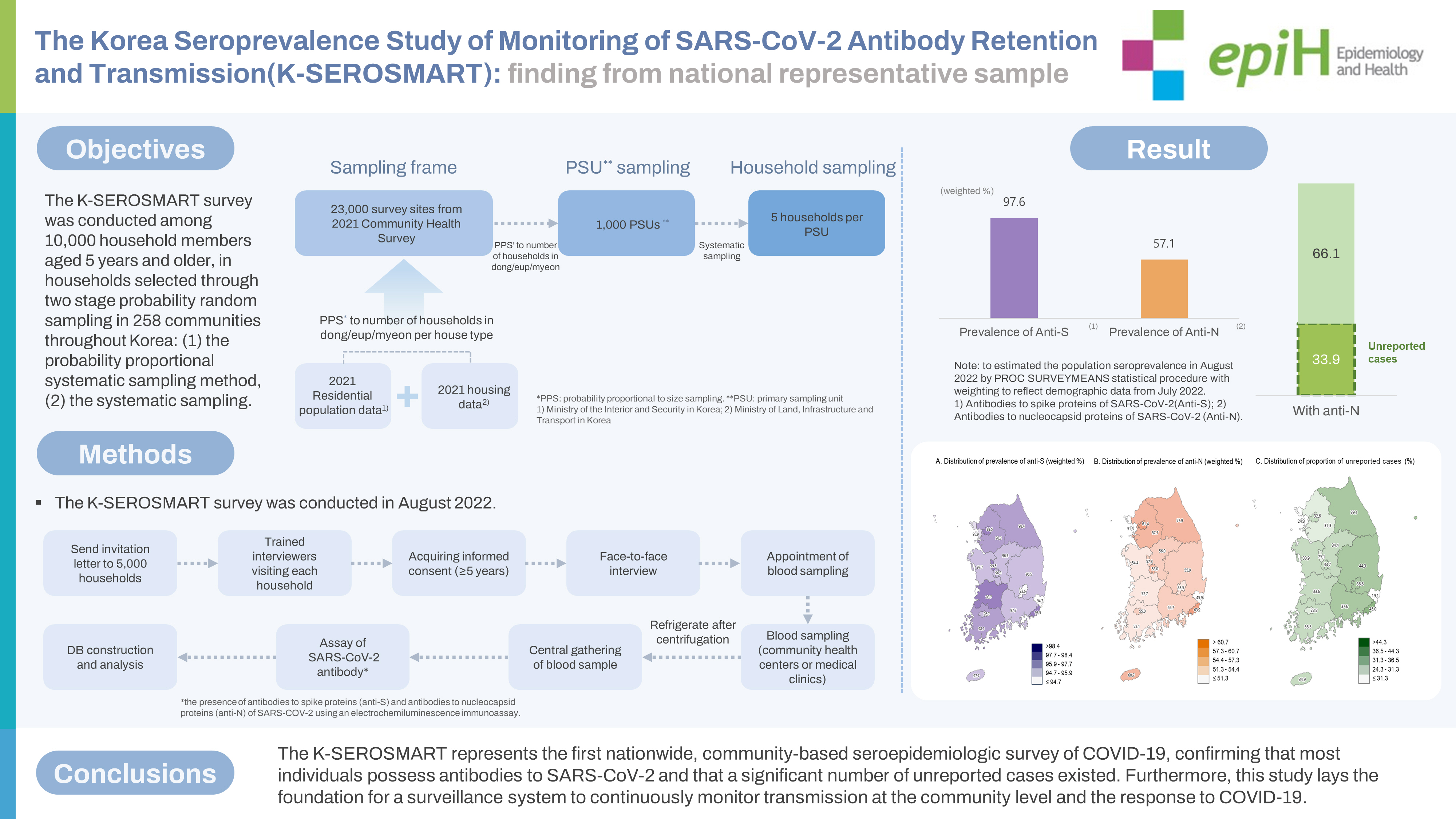
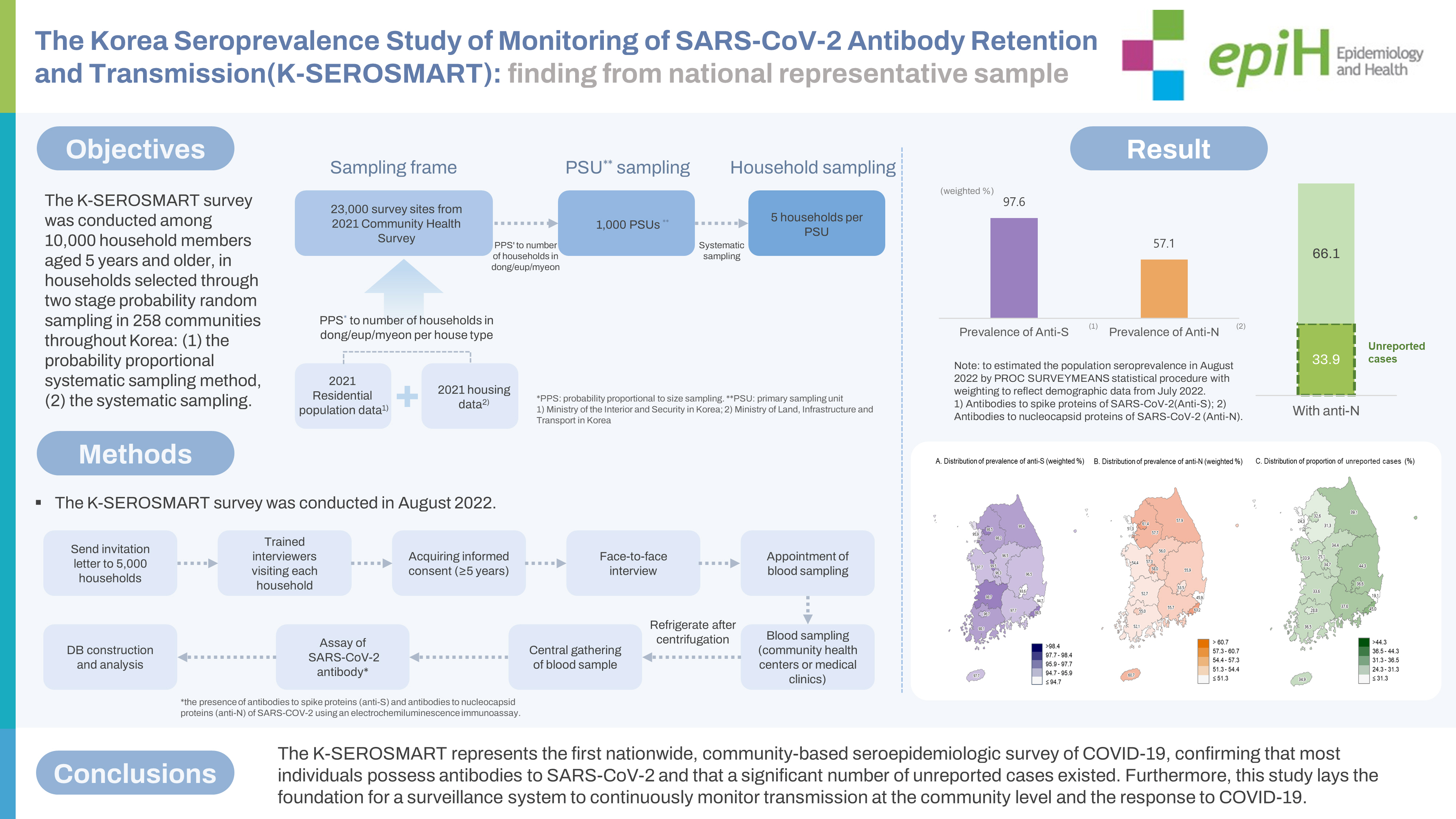
- We estimated the population prevalence of antibodies to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), including unreported infections, through a Korea Seroprevalence Study of Monitoring of SARS-CoV-2 Antibody Retention and Transmission (K-SEROSMART) in 258 communities throughout Korea.
-
METHODS
- In August 2022, a survey was conducted among 10,000 household members aged 5 years and older, in households selected through two stage probability random sampling. During face-to-face household interviews, participants self-reported their health status, COVID-19 diagnosis and vaccination history, and general characteristics. Subsequently, participants visited a community health center or medical clinic for blood sampling. Blood samples were analyzed for the presence of antibodies to spike proteins (anti-S) and antibodies to nucleocapsid proteins (anti-N) SARS-CoV-2 proteins using an electrochemiluminescence immunoassay. To estimate the population prevalence, the PROC SURVEYMEANS statistical procedure was employed, with weighting to reflect demographic data from July 2022.
-
RESULTS
- In total, 9,945 individuals from 5,041 households were surveyed across 258 communities, representing all basic local governments in Korea. The overall population-adjusted prevalence rates of anti-S and anti-N were 97.6% and 57.1%, respectively. Since the Korea Disease Control and Prevention Agency has reported a cumulative incidence of confirmed cases of 37.8% through July 31, 2022, the proportion of unreported infections among all COVID-19 infection was suggested to be 33.9%.
-
CONCLUSIONS
- The K-SEROSMART represents the first nationwide, community-based seroepidemiologic survey of COVID-19, confirming that most individuals possess antibodies to SARS-CoV-2 and that a significant number of unreported cases existed. Furthermore, this study lays the foundation for a surveillance system to continuously monitor transmission at the community level and the response to COVID-19.
- On March 11, 2020, the World Health Organization declared coronavirus disease 2019 (COVID-19) a global pandemic [1]. As of July 31, 2022, over 580 million confirmed cases of COVID-19 have been reported worldwide, resulting in approximately 6.42 million deaths. In Korea, community outbreak has persisted since the emergence of COVID-19 (Figure 1), with over 19.77 million confirmed cases and more than 25,000 deaths reported [2].
- In Korea, the test-trace-isolate-quarantine (TTIQ) containment strategy has been employed since the beginning of the COVID-19 pandemic in 2020 to promptly identify infected individuals and disrupt transmission routes through rapid testing and extensive follow-up [3]. However, in late 2020, the third peak of COVID-19 occurred. From then onward, the proportion of cases with epidemiologic transmission routes unknown at the time of diagnosis increased, resulting in many unreported cases within the community [4]. These cases have been reported to account for 5% to 80% of individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in a systematic review and meta-analysis worldwide [5], who shed the virus at similar levels comparable to symptomatic individuals [6,7]. Although they do not require medical care, these cases contribute to the spread of the disease within the community [8].
- The World Health Organization recommends population-based seroepidemiologic surveys to determine the prevalence of antibodies to SARS-CoV-2, including asymptomatic infections in the community [9]. Seroepidemiologic surveys of COVID-19 can be used to monitor and assess the level of immunity to the virus in a population by directly measuring the presence of antibodies resulting from natural infection or vaccination [10-12].
- Serotracking data reveals that as of November 2022, a total of 3,924 seroprevalence surveys for COVID-19 have been carried out in 140 countries worldwide [13]. A meta-analysis based on some of these serotracker data, excluding studies with incorrect sample frames and methodological shortcomings, highlights that population-based estimates of COVID-19 far exceed reported cases. The study emphasizes the importance of conducting high-quality and standardized collaborative serosurveillance through random sampling to monitor the COVID-19 pandemic and prepare for potential emerging viruses in the future [14]. The United Kingdom adopted a random sampling approach, surveying 150,000 people every month for 23 months starting from May 2020, during the early phase of the pandemic. The Real-time Assessment of Community Transmission Study produced community-based data on antibody-positive rates repeatedly. This data serves as the foundation for understanding the spread of the pandemic and establishing effective quarantine response strategies [15].
- The COVID-19 Serosurveillance Network in Australia routinely calculates the seroprevalence of COVID-19 using residual blood from donors aged 18 years and older [16]. In the United States, the prevalence of anti-nucleocapsid proteins of SARS-CoV-2 was reported every 4 weeks from September 2021 to April 2022 using blood samples collected at healthcare facilities for clinical trials [17]. Additionally, extra data on the seroepidemiology of COVID-19 were collected in the 2021-2022 National Health and Nutrition Examination Survey to overcome the limitations of convenience sampling [18].
- In Korea, several seroprevalence surveys for COVID-19 have been conducted; however, most of these studies were small-scale and relied on convenience sampling. Furthermore, these surveys did not provide analysis by region, age, or other socio-demographic factors. The use of residual sample from other survey systems has been limited in calculating a representative prevalence of antibodies to SARS-CoV-2 [4,12,19,20]. The prevalence of antibodies to SARS-CoV-2 may vary based on factors such as the survey population, sampling method, timing and location of the survey, and blood sample analysis technique employed [11].
- To accurately estimate the scope of COVID-19 transmission, a nationwide survey of a representative sample of the population is necessary. In Korea, the Community Health Survey (CHS) has been conducted since 2008. This survey, utilizing a regionally representative sampling and survey system, is a collaborative effort between the Korea Disease Control and Prevention Agency (KDCA), 17 provinces, 258 community health centers, and 34 universities [21,22]. The Korea Seroprevalence Study of Monitoring of SARS-CoV-2 Antibody Retention and Transmission (K-SEROSMART) aimed to conduct a national seroepidemiologic survey of COVID-19 using the CHS system to estimate the population prevalence of antibodies to SARS-CoV-2.
INTRODUCTION
- System for the community-based seroepidemiologic survey of COVID-19
- The Korean Society of Epidemiology, which supervised the survey in collaboration with the National Institute of Health of the KDCA, organized 4 teams: sampling design, field survey, specimen analysis, and data analysis teams. The sampling design team selected and managed the primary sampling units (PSUs) to ensure sample representativeness and calculated population-adjusted weights. The field survey team carried out household surveys and collected blood samples. The specimen analysis team transported blood samples to central lab and performed laboratory tests for SARS-CoV-2 antibodies, while the data analysis team compiled the survey and laboratory test results into a database and performed analyses. Furthermore, a multidisciplinary research committee was established to monitor the survey process and make key decisions.
- In this study, the field survey team employed a CHS consortium [22], which included 34 universities, 258 community health centers, and 132 community medical clinics. The participating universities coordinated the survey participants, interviewers, and blood collection sites (community health centers or medical clinics) within each region, while also monitoring the survey process and ensuring quality control. Community health centers and medical centers were responsible for monitoring the status of blood collection in their respective regions and carrying out the blood collection procedures (Supplementary Material 1).
- The CHS was initiated by the Korea Centers for Disease Control and Prevention in 2008 under the Community Health Act (Article 4). It is a community-based cross-sectional survey conducted among local residents aged 19 years and over who live within the jurisdictions of community health centers. Each year, approximately 900 individuals from each community and 230,000 in total nationwide were surveyed through face-to-face interviews. Furthermore, health statistics on health behaviors and levels of chronic disease at the regional level were generated. For representative sampling, PSUs were selected using probability proportional systematic sampling stratified by dong (neighborhood), eup (town), myeon (township), and housing type, and sample households were selected through systematic sampling. A consortium of 258 community health centers and 34 universities across the country was formed to standardize the survey procedures and implementation system, as well as to establish a sustainable community survey monitoring system by creating a network within the community. The statistical data generated are used for healthcare planning and to evaluate the performance of health projects in the regions of Korea.
- Sample selection
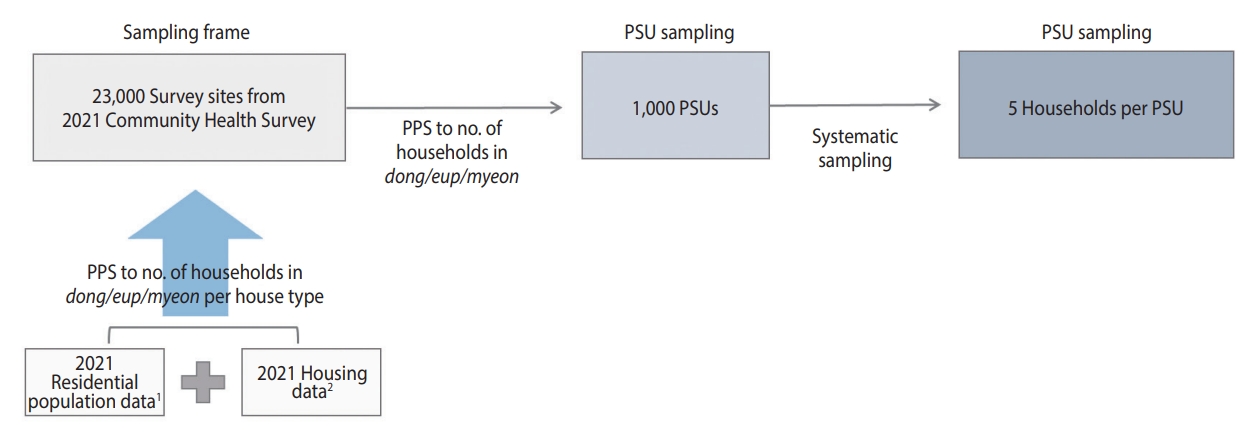
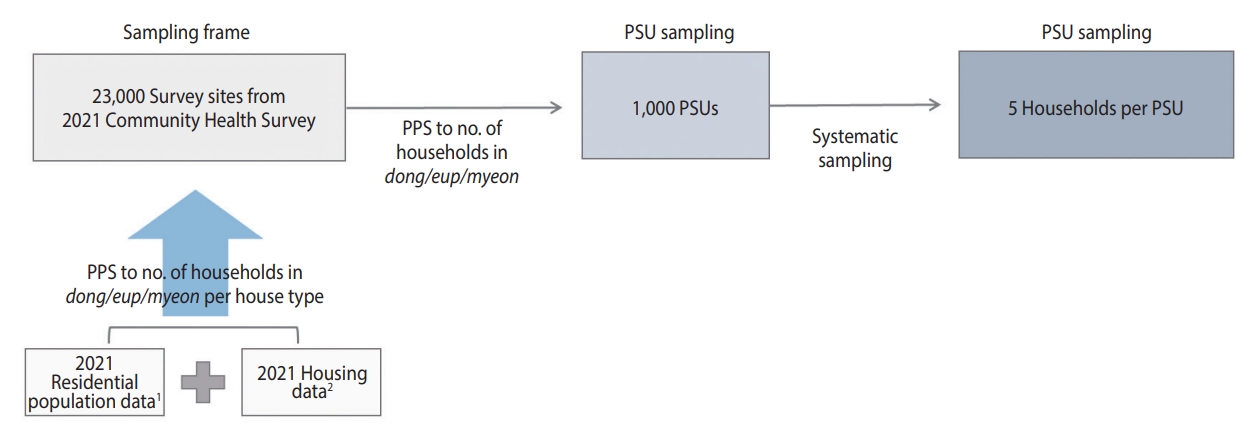
- The sample size was set at 10,000 to ensure the representativeness of the prevalence of antibodies to SARS-CoV-2 [23]. The survey population consisted of residents aged 5 years and older, residing in residential housing (apartments or houses) within each PSU at the time of the survey, and who were eligible for blood sampling.
- The sampling design process is illustrated in Figure 2. The sampling frame for selecting the PSUs was based on 23,000 survey sites from the 2021 CHS, from which 1,000 PSUs were chosen. The PSUs selected from the 2021 CHS were determined using the probability proportional systematic sampling method. This method considered the number of sample points allocated to each dong, eup, and myeon, based on the number of households by housing type in the sampling frame. This frame linked the residential population data from the Ministry of the Interior and Safety with the housing data from the Ministry of Land, Infrastructure, and Transport from 2021 [21,22,24].
- The 1,000 PSUs were allocated proportionally, considering the number of population by each si, gun, gu, representing all basic local governments. To ensure the participation of all 258 community health centers, 2 PSUs per health center were initially allocated, and the remaining points were distributed based on the population aged 5 years and older in July 2022 for each city, county, and district (Table 1).
- In the sampling frame, the number of PSUs allocated to each city, county, and district was selected using the probability proportional systematic sampling method, based on the number of households in tong (urban village), ban (hamlet), and ri (rural village). To account for participant access to blood collection sites, we have excluded PSUs located in isolated areas. In contrast, only 1 health center is in Sejong; however, 10 PSUs were selected to ensure the representativeness of the sample for comparison among provinces.
- To choose the sample households for each PSU, we employed the systematic sampling technique. This involved identifying the total number of households within each PSU and selecting 5 primary households along with 2 backup households for each primary household. The reserve households were chosen based on the numbers subsequent to the serial number corresponding to the primary household.
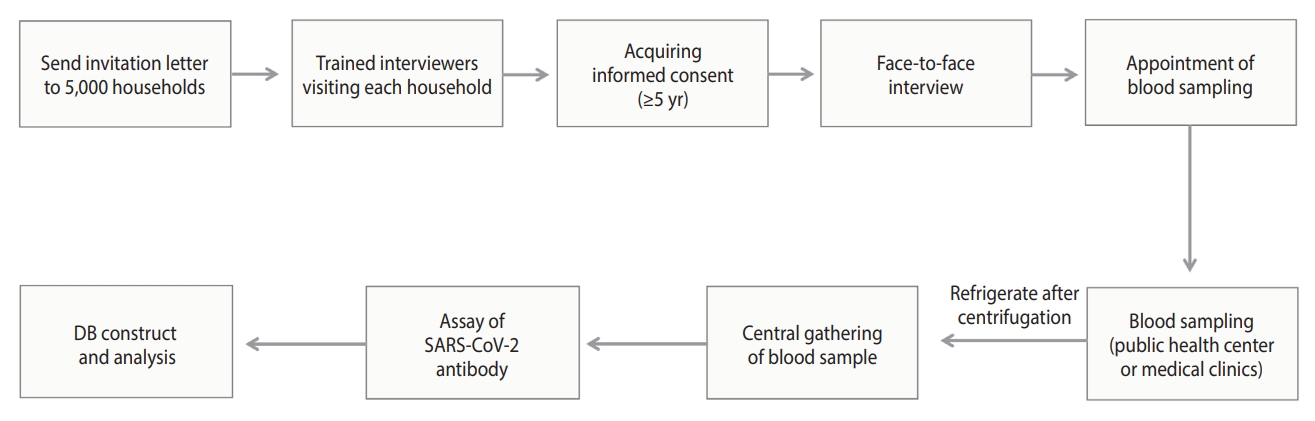
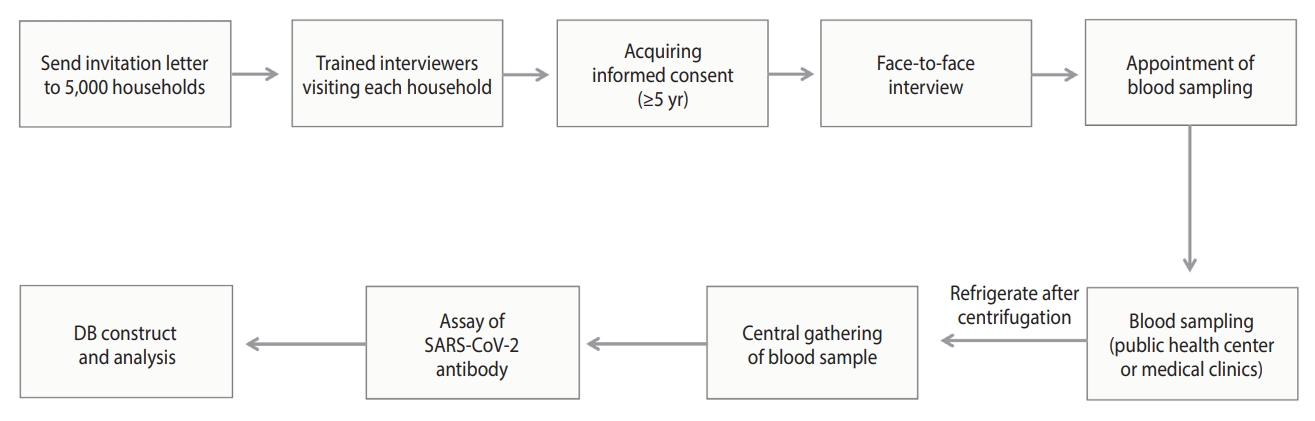
- Survey process
- The survey process is illustrated in Figure 3. Households chosen for the survey received a letter informing them of their eligibility for the study. Trained interviewers visited these households to obtain consent from household members aged 5 years and older. For individuals under the age of 18 years, consent was obtained from a legal representative. The survey was conducted through face-to-face interviews, during which participants self-reported information about their health status, disease history, COVID-19 diagnosis and vaccination history, and socioeconomic status (education, household income, and generational household) to the interviewer. After completing the interview, participants followed the interviewer’s instructions to schedule a time and location for a blood draw.
- Blood samples were collected at the community health centers located in the participants’ residential areas, with children aged 5 years to 18 years advised to have their samples collected at designated medical clinics. In accordance with the blood collection protocol, 4 mL of blood was collected from adults and 2 mL from children. The samples were then centrifuged for 10 minutes at 3,000 rpm and subsequently refrigerated.
- To ensure the representativeness of the sample, a minimum of 3 households or 10 individuals were surveyed per PSU. Households that either refused to participate or were ineligible for the survey (e.g., those composed of non-resident adults, foreign residents, household members with COVID-19-related symptoms or under quarantine, and those unable to travel for blood collection) were replaced with a designated reserve household. Furthermore, if the target number of participants for each area was not met, additional visits were made to reserve households.
- Laboratory testing
- Blood samples were gathered from the community health centers and designated medical clinics, then brought to regional laboratory centers on the same day. These samples were stored according to provided guidelines and transported to the central laboratory for analysis the following morning. Antibody testing for SARS-CoV-2 was conducted using the Elecsys Anti-SARS-CoV-2 reagent (Roche, Mannheim, Germany) in accordance with the manufacturer’s protocol. The testing was performed on a Cobas e801 analyzer (Roche), utilizing the electrochemiluminescence immunoassay principle. The clinical sensitivity and specificity of this test were 99.5% and 99.8%, respectively [4,25].
- Spike proteins and nucleocapsid proteins serve as the primary antigens for detecting antibodies to SARS-CoV-2. The spike protein enables the virus to interact with human cells, while the nucleocapsid protein, as the most abundant protein in the coronavirus, is believed to play a role in viral particle assembly and release [26]. Antibodies to spike proteins (anti-S) and antibodies to nucleocapsid proteins (anti-N) are utilized as indicators of an effective immune response including vaccine effectiveness and as biomarkers of natural infection, respectively [27].
- The results of the anti-S assay were presented numerically and deemed positive for anti-S when the cut-off index was greater than or equal to 0.80 U/mL, signifying the formation of antibodies due to natural infection or vaccination [28]. The results of the anti-N assay were classified as reactive or non-reactive, with a specified cut-off index. A cut-off index of 1.0 or higher indicated an anti-N result of reactive, meaning that the patient tested positive for anti-N; this suggested that antibodies had previously formed due to natural infection [28]. Importantly, the degree to which the cut-off index for the anti-N assay is higher than 1.0 does not represent the total quantity of antibodies present in the sample.
- Statistical analysis
- To estimate the population prevalence of anti-S and anti-N, we employed the PROC SURVEYMEANS procedure in SAS version 9.4 (SAS Institute Inc., Cary, NC, USA), utilizing sample weights based on the July 2022 registered population [22-24].
- The sample weight was calculated by multiplying 3 factors: (1) sampling design weight, (2) non-response correction weight, and (3) post-stratification correction weight. (i) Sampling design weight is the reciprocal of the sampling rate, which was determined by dividing the number of people assigned to a health center unit by the number of registered residents. (ii) Non-response correction weight was calculated as the inverse of the response rate by health center unit, under the assumption that the characteristics of non-respondents and respondents were similar. (iii) Post-stratification adjustment weights were used to equalize the distribution of the sample by sex and age group, both of which influence antibody prevalence, with the distribution of the population. These weights were calculated by dividing the number of residents by sex and age group at each site by the sum of the design weight and the non-response adjustment weight.
- The prevalence of anti-S and anti-N is presented with 95% confidence intervals (CIs) and relative standard error (RSE). The RSE was calculated by dividing the standard error by the corresponding estimate and expressing it as a percentage (%). In household surveys, an RSE of 25% or lower is considered to indicate high confidence in the estimated value [29].
- In this study, the difference between the anti-N prevalence and the cumulative COVID-19 incidence reported by the KDCA was defined as the unreported infection rate [4]. To compare the differences in cumulative incidence by sex, age, and provinces, the anti-N prevalence was further calculated using age-standardized weighting, with the December 2021 population serving as the reference.
- The cumulative incidence of COVID-19 was determined using the total number of confirmed cases reported to the Korean COVID-19 case reporting system between January 20, 2020 and July 31, 2022 as the numerator, and the resident population in December 2021 as the denominator.
- The reporting system for confirmed COVID-19 cases in Korea operates as follows. Medical institutions are required to report a confirmed case (either a positive professional rapid antigen test or an emergency screening test result) to the community health center within 24 hours of initially identifying the case, then enter the case report into the COVID-19 Information Management System. Community health centers must then report the case to the city health department within 24 hours of initially identifying the case (upon confirmation of a positive result from a health and environmental laboratory or testing contractor) and enter the outbreak report into the COVID-19 Information Management System [30].
- Ethics statement
- This survey received an exemption from review by the Institutional Review Board of the KDCA, in accordance with Article 36 of the Bioethics Act, Article 33 of the Enforcement Rules, and Article 2 of the Bioethics Act. The study was deemed to require urgent action for public health and was conducted directly or commissioned by the state or local government to review and evaluate public welfare or service programs (2022-07-04-PE-A).
MATERIALS AND METHODS
- The face-to-face interviews took place from August 11, 2022 to August 30, 2022, while blood samples were collected between August 12, 2022 and September 5, 2022. In total, 9,945 individuals from 5,041 households participated in the survey. These participants included 4,008 individuals from 1,957 primary households and 5,938 individuals from 3,084 backup households. The prevalence of anti-S and anti-N, based on various characteristics, is presented in Table 2.
- The prevalence of anti-S was 97.6% (95% CI, 97.2 to 97.9). By age group, the anti-S prevalence was lowest among 5-9-year-olds at 81.5% (95% CI, 76.5 to 86.4) and increased with age, reaching a peak of 99.4% (95% CI, 98.9 to 99.8) in the 70-79-year group. Significant differences were also found based on household income and generational household. In terms of provinces, the prevalence of anti-S proteins was lowest in Daegu at 93.6% (95% CI, 90.8 to 96.3) and highest in Sejong at 99.5% (95% CI, 98.4 to 100.0), with statistically significant differences observed between provinces (Figure 4A).
- The prevalence of anti-N was found to be 57.1% (95% CI, 56.0 to 58.2), with rates of 56.4% (95% CI, 54.7 to 58.1) in male and 57.9% (95% CI, 56.4 to 59.3) in female. By age group, the highest prevalence of anti-N proteins was observed in 5-9-year-olds at 82.1% (95% CI, 77.3 to 87.0), and it decreased with age, reaching its lowest point of 31.2% (95% CI, 27.2 to 35.2) in individuals aged 80 years and older. The anti-N prevalence increased with education and household income, with the lowest prevalence of 44.6% (95% CI, 41.3 to 47.9) found in single-person households and the highest prevalence of 60.8% (95% CI, 59.2 to 62.4) in second generation. By province, the lowest prevalence of anti-N was found in Ulsan at 45.9% (95% CI, 37.7 to 54.1) and the highest in Busan at 63.2% (95% CI, 59.3 to 67.2), with statistically significant differences between provinces (Figure 4B).
- The proportion of unreported infections was 33.9% overall (36.6% in male and 30.5% in female participants). Regarding age group, the highest proportions were observed in those 50-59 years, 60-69 years, and 70-79 years old, at 47.9%, 45.0%, and 40.5%, respectively, while the lowest proportion was found in those 80 years and older, at 12.9% (Table 3). By metropolitan area, the lowest proportion in Ulsan (19.1%) and the highest in Busan (45.0%), constituting a 2.4-fold difference in the proportion of unreported infections between the regions (Figure 4C).
RESULTS
- In this study, we carried out a seroepidemiologic survey of COVID-19 among 9,945 residents aged 5 years and older from August 11, 2022 to September 5, 2022, several months after the surge of COVID-19 cases mainly due to Omicron variants in early 2022 in Korea. The K-SEROSMART was based on the CHS system and was designed to determine the representative population prevalence of anti-S and anti-N in Korea.
- The prevalence of anti-S was 97.6%, indicating that most Koreans possess anti-S. No significant differences were observed based on sex or region; however, the prevalence of anti-S was lower among 5-9-year-olds (81.5%) compared to other age groups. As of August 2022 in Korea, despite recommendations of at least 1 dose for children aged 12-17 years and high-risk children aged 5-11 years [31], the vaccination rate for 5-11-year-olds was only 1.5% [32]. Furthermore, the prevalence of anti-N among 5-9-year-olds was 82.1%, similar to the prevalence of anti-S proteins, suggesting that the anti-S produced in this age group is probably due to natural infection.
- The anti-N prevalence rate was 57.1%. No significant differences were observed by sex; however, the prevalence of anti-N proteins decreased with increasing age. In another study, the prevalence of anti-N, as estimated from 5,005 blood samples collected between August 23, 2022 and September 2, 2022, in Australia, was 65.2% [16], Additionally, the prevalence from 13,787 blood samples collected between June 29, 2022 and July 19, 2022, in the United Kingdom was 73.4% [33]. This could be interpreted either as a success in preventing the spread of COVID-19 in Korea or as an indication of the potential for COVID-19 to spread [34].
- Since the onset of the COVID-19 outbreak, Korea has implemented a swift and comprehensive response system, involving numerous organizations alongside the TTIQ strategy. Furthermore, its citizens have actively participated in preventative measures such as wearing masks, practicing social distancing, and becoming vaccinated. Consequently, the scale of COVID-19 infections in Korea has been much smaller than in other countries.
- Evidence indicates that COVID-19 vaccination reduces the incidence, severity, hospitalization, and mortality of COVID-19 [35]. In Korea, individuals aged 50 years and older have been prioritized for COVID-19 vaccination. Consequently, as of July 2022, the vaccination rate for this age group reached 96.7%, and the booster update rate was 87.0%, with vaccination rates increasing with age [36]. This finding suggests that the prevalence of anti-N positivity decreases with age, possibly indicating the effectiveness of the vaccine. However, the lower anti-N prevalence in older age groups and among those with comorbidities implies that they may still be at risk of natural infection. These individuals could potentially become a high-risk patient population in the event of an outbreak and should be considered for ongoing monitoring and response measures.
- In Korea, the proportion of unreported infections was 33.9%. This represents a significant increase from the 18.3% [37] of unreported infections reported in the residual blood testing of participants in the National Health and Nutrition Examination Survey in April 2022. The likely cause of this increase is the rise in the number of unreported infections due to the Omicron pandemic. However, this figure is lower than the 43.7% of unreported infections among adults aged 18 years and older reported in the 2021-2022 National Health and Nutrition Examination Survey in the United States [18]. This difference may be attributed to the extensive follow-up of patient contacts under the TTIQ policy in the early stages of the COVID-19 outbreak, as previously described [3].
- Following the Omicron surge in early 2022, the TTIQ policy was relaxed, which may have further increased the proportion of hidden COVID-19 cases in the community. The proportion of unreported infections is an important measure of the extent of COVID-19 transmission within a community [38-40]. In the present study, the proportion of unreported infections was higher among male participants and those aged 50-59 years and 60-69 years, and it varied by up to a factor of approximately 2.4 across provinces. The impact of the COVID-19 pandemic was disproportionately influenced by individual socioeconomic characteristics, and studies have reported differences in response capacity and policy compliance [41,42]. Further investigation and in-depth analysis of socioeconomic factors such as occupation and employment type that may influence differences in the proportion of unreported infections, as well as COVID-19 policies in each region, are needed to develop complementary policies that consider socioeconomic characteristics.
- By utilizing an existing consortium of community health centers and universities [21] and forming a collaborative network with regional medical clinics, the present survey efficiently constituted a systematic, rapid, and cost-effective seroepidemiologic investigation of COVID-19. This will provide a foundation for future monitoring of COVID-19 antibodies by tracking existing participants to observe changes in antibody levels. Additionally, it will persist in facilitating observation of the spread of and response to COVID-19 within the community, while also functioning as a surveillance system for other emerging infectious diseases.
- Limitations of this study include the following. First, the prevalence of COVID-19 antibodies may be underestimated in some or all populations. This community-based cross-sectional survey may be subject to survivorship bias, as deaths or certain hospitalizations due to COVID-19 (particularly common among older adults) would have prevented inclusion. Furthermore, even in cases of natural infection, antibody formation may not have occurred or antibody titers could have been lost over time; this depends on factors such as the level of pathogen exposure, individual susceptibility to disease, timing of vaccination and natural infection, underlying medical conditions, and the presence of breakthrough infections [17,43-45]. Second, the participation rate of children and young people, who had difficulty in visiting community health centers or medical clinics for blood sampling, was low. However, the parameters were estimated by weighting under the assumption that no difference existed in antibody prevalence based on survey participation. Third, we used the cumulative number of COVID-19 cases reported by the KDCA to estimate the scale of unreported infections in the community. Since the diagnosis of COVID-19 was only available for patients who visited a medical institution or health center, the rate of unreported infection in the community may be underestimated.
- In August 2022, a nationwide community-based seroepidemiologic survey for COVID-19 revealed that most of the Korean population had acquired anti-S through vaccination or natural infection, and that 1 in 3 naturally infected individuals had an unreported infection. This survey is the first of its kind in Korea to determine a representative COVID-19 antibody prevalence rate by systematically and cost-effectively surveying many people within a short period (about 3 weeks). This was achieved through collaboration with community health centers, universities, and medical clinics in the region. The survey is also noteworthy for establishing the foundation of a surveillance system that can be used to continuously monitor the spread of COVID-19 infection and regional responses. In addition to follow-up and periodic surveys of the participants, the data will be used as a basis for developing COVID-19 prevention policies. This will be accomplished by identifying the characteristics of COVID-19 spread in the community, factors influencing undetected infections, and fluctuations in antibody titers through linkage with various sources. These sources include COVID-19 confirmation information and vaccination history from the KDCA, as well as morbidity information from the Korea Health Insurance Corporation.
DISCUSSION
SUPPLEMENTARY MATERIALS
-
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare for this study.
-
FUNDING
This study was supported by a grant from the National Institutes of Health of the Korea Disease Control and Prevention Agency (2022-ER2603-00).
-
AUTHOR CONTRIBUTIONS
Conceptualization: Kwon J, Kim SS, Kim B, Lee JW, Jang EY, Kim AR, Nam J, Kim DH, Lee SY. Data curation: Han J, Baek HJ, Noh E, Yoon K, Lee KO. Formal analysis: Han J, Noh E. Funding acquisition: Kim DH, Kwon J, Kim SS, Kim B, Lee JW, Jang EY, Kim AR, Nam J. Methodology: Han J, Baek HJ, Noh E, Yoon K, Kim JA, Ryu S, Lee KO, Park NY, Jung E, Kim S, Lee H, Hwang YS, Jung J, Lee HJ, Cho S, Oh S, Kim M. Project administration: Kim B, Lee JW, Jang EY, Kim AR, Nam J, Kim DH, Lee SY, Park NY, Kim S, Jung J, Lee HJ, Cho S, Oh CM, Yu B, Hong YS, Kim K, Jung S, Han MA, Lee MS, Lee JJ, Hwangbo Y, Yim HW, Kim YM, Lee J, Lee WY, Park JH, Oh S, Jo HS, Kim H, Kang G, Nam HS, Lee JH, Oh GJ, Shin MH, Ryu S, Hwang TY, Park SW, Kim SK, Seol R, Park KS, Kim SY. Visualization: Han J, Baek HJ, Noh E. Writing – original draft: Kim DH, Lee SY, Han J, Baek HJ, Noh E, Yoon K, Kim JA, Ryu S, Oh CM, Yu B, Hong YS, Kim K, Jung S, Han MA, Lee MS, Lee JJ, Hwangbo Y, Yim HW, Kim YM, Lee J, Lee WY, Park JH, Oh S, Jo HS, Kim H, Kang G, Nam HS, Lee JH, Oh GJ, Shin MH, Ryu S, Hwang TY, Park SW, Kim SK, Seol R, Park KS, Kim SY. Writing – review & editing: Kim DH, Lee SY, Han J, Baek HJ, Lee KO, Park NY, Jung E, Kim S, Lee H, Hwang YS, Jung J, Lee HJ, Cho S, Oh S, Kim M, Kwon J, Kim SS, Kim B, Lee JW, Jang EY, Kim AR, Nam J
NOTES
ACKNOWLEDGEMENTS


| Characteristics | n |
Prevalence of anti-S |
Prevalence of anti-N |
|||||
|---|---|---|---|---|---|---|---|---|
| Weighted % (95% CI) | RSE | p-value1 | Weighted % (95% CI) | RSE | p-value1 | |||
| Overall | 9,945 | 97.6 (97.2, 97.9) | 0.2 | - | 57.1 (56.0, 58.2) | 1.0 | - | |
| Sex | Male | 4,474 | 97.2 (96.6, 97.7) | 0.3 | 0.024 | 56.4 (54.7, 58.1) | 1.5 | 0.187 |
| Female | 5,471 | 98.0 (97.5, 98.4) | 0.2 | 57.9 (56.4, 59.3) | 1.3 | |||
| Age (yr) | 5-9 | 297 | 81.5 (76.5, 86.4) | 3.1 | <0.001 | 82.1 (77.3, 87.0) | 3.0 | <0.001 |
| 10-19 | 757 | 92.9 (90.9, 94.8) | 1.1 | 69.3 (65.8, 72.8) | 2.6 | |||
| 20-29 | 887 | 98.8 (98.1, 99.6) | 0.4 | 60.7 (57.1, 64.2) | 3.0 | |||
| 30-39 | 934 | 98.4 (97.5, 99.3) | 0.5 | 61.5 (58.1, 64.9) | 2.8 | |||
| 40-49 | 1,337 | 98.6 (97.9, 99.3) | 0.4 | 59.7 (56.8, 62.6) | 2.5 | |||
| 50-59 | 1,660 | 99.2 (98.7, 99.7) | 0.2 | 54.5 (51.9, 57.2) | 2.5 | |||
| 60-69 | 1,994 | 99.2 (98.8, 99.7) | 0.2 | 50.4 (47.9, 52.8) | 2.5 | |||
| 70-79 | 1,450 | 99.4 (98.9, 99.8) | 0.2 | 43.0 (40.2, 45.8) | 3.3 | |||
| ≥80 | 629 | 97.3 (95.8, 98.8) | 0.8 | 31.2 (27.2, 35.2) | 6.5 | |||
| Education (age ≥19 yr) | Primary school | 1,555 | 99.1 (98.6, 99.6) | 0.3 | 0.721 | 40.1 (37.4, 42.9) | 3.4 | <0.001 |
| Middle/High school | 3,702 | 98.9 (98.5, 99.3) | 0.2 | 54.4 (52.5, 56.2) | 1.7 | |||
| Postsecondary | 3,582 | 98.7 (98.3, 99.2) | 0.2 | 58.2 (56.4, 60.0) | 1.6 | |||
| Household income (/103 KRW) | <2,000 | 1,940 | 97.8 (97.0, 98.7) | 0.5 | 0.007 | 43.1 (40.5, 45.7) | 3.1 | <0.001 |
| 2,000-3,999 | 2,391 | 98.3 (97.7, 98.9) | 0.3 | 56.7 (54.4, 59.0) | 2.1 | |||
| 4,000-5,999 | 1,369 | 96.4 (95.3, 97.5) | 0.6 | 59.0 (56.1, 62.0) | 2.5 | |||
| 6,000-7,999 | 1,565 | 96.7 (95.6, 97.7) | 0.5 | 57.4 (54.7, 60.2) | 2.5 | |||
| ≥8,000 | 1,288 | 97.9 (97.0, 98.8) | 0.5 | 64.7 (61.9, 67.6) | 2.3 | |||
| Generational household | Single person | 1,234 | 98.4 (97.5, 99.3) | 0.5 | <0.001 | 44.6 (41.3, 47.9) | 3.8 | <0.001 |
| First generation | 2,802 | 99.3 (99.0, 99.7) | 0.2 | 52.3 (50.1, 54.4) | 2.1 | |||
| Second generation | 4,195 | 96.4 (95.8, 97.1) | 0.3 | 60.8 (59.2, 62.4) | 1.4 | |||
| Third generation | 526 | 97.6 (96.0, 99.2) | 0.9 | 58.4 (53.6, 63.2) | 4.2 | |||
| Province | Seoul | 1,399 | 98.5 (97.8, 99.2) | 0.3 | <0.001 | 61.4 (58.7, 64.2) | 2.3 | <0.001 |
| Busan | 636 | 98.5 (97.6, 99.4) | 0.5 | 63.2 (59.3, 67.2) | 3.2 | |||
| Daegu | 382 | 93.6 (90.8, 96.3) | 1.5 | 53.5 (48.2, 58.8) | 5.1 | |||
| Incheon | 485 | 95.9 (94.1, 97.8) | 1.0 | 51.3 (46.0, 56.6) | 5.3 | |||
| Gwangju | 228 | 96.7 (94.2, 99.3) | 1.3 | 55.0 (47.8, 62.2) | 6.7 | |||
| Daejeon | 230 | 96.3 (93.7, 98.9) | 1.4 | 58.0 (50.9, 65.1) | 6.3 | |||
| Ulsan | 192 | 94.7 (91.0, 98.3) | 2.0 | 45.9 (37.7, 54.1) | 9.1 | |||
| Sejong | 100 | 99.5 (98.4, 100.0) | 0.5 | 57.3 (47.4, 67.1) | 8.8 | |||
| Gyeonggi | 2,173 | 98.2 (97.6, 98.8) | 0.3 | 57.7 (55.5, 59.9) | 2.0 | |||
| Gangwon | 495 | 98.4 (97.1, 99.8) | 0.7 | 57.9 (53.3, 62.6) | 4.1 | |||
| Chungbuk | 431 | 96.5 (94.4, 98.6) | 1.1 | 56.0 (50.4, 61.6) | 5.1 | |||
| Chungnam | 513 | 97.7 (96.0, 99.4) | 0.9 | 54.4 (49.3, 59.5) | 4.8 | |||
| Jeonbuk | 440 | 98.7 (97.3, 100.0) | 0.7 | 52.7 (47.0, 58.5) | 5.5 | |||
| Jeonnam | 611 | 98.1 (96.7, 99.4) | 0.7 | 52.1 (47.8, 56.4) | 4.2 | |||
| Gyeongbuk | 738 | 96.5 (94.8, 98.2) | 0.9 | 55.9 (51.8, 59.9) | 3.7 | |||
| Gyeongnam | 710 | 97.7 (96.2, 99.1) | 0.8 | 55.7 (51.4, 60.1) | 4.0 | |||
| Jeju | 182 | 97.7 (94.4, 100.0) | 1.7 | 60.7 (53.1, 68.4) | 6.4 | |||
Seroprevalence was estimated with sampling weights using the population of registered residents in July 2022.
SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; CI, confidence interval; RSE, relative standard error; anti-S, antibodies to spike proteins; anti-N, antibodies to nucleocapsid proteins; KRW, Korean won.
1 From the Rao-Scott chi-square test.
| Characteristics | Age-standardized prevalence rate of anti-N1 (A) | Cumulative rate of confirmed COVID-19 reported by KCDC2 (B) | Proportion of unreported infections ([A−B]/A) | |
|---|---|---|---|---|
| Overall | 57.2 | 37.8 | 33.9 | |
| Sex | ||||
| Male | 56.5 | 35.8 | 36.6 | |
| Female | 58.0 | 40.3 | 30.5 | |
| Age (yr) | ||||
| 5-9 | 82.2 | 66.6 | 19.0 | |
| 10-19 | 69.5 | 55.3 | 20.4 | |
| 20-29 | 60.3 | 44.2 | 26.7 | |
| 30-39 | 61.7 | 43.4 | 29.7 | |
| 40-49 | 59.7 | 37.3 | 37.5 | |
| 50-59 | 54.5 | 28.4 | 47.9 | |
| 60-69 | 50.4 | 27.7 | 45.0 | |
| 70-79 | 42.7 | 25.4 | 40.5 | |
| ≥80 | 30.9 | 26.9 | 12.9 | |
| Province | ||||
| Seoul | 61.6 | 41.5 | 32.6 | |
| Busan | 63.6 | 35.0 | 45.0 | |
| Daegu | 53.5 | 33.9 | 36.6 | |
| Incheon | 51.1 | 38.7 | 24.3 | |
| Gwangju | 54.6 | 38.9 | 28.8 | |
| Daejeon | 57.9 | 37.8 | 34.7 | |
| Ulsan | 46.1 | 37.3 | 19.1 | |
| Sejong | 53.7 | 40.2 | 25.1 | |
| Gyeonggi | 56.9 | 39.1 | 31.3 | |
| Gangwon | 59.6 | 36.3 | 39.1 | |
| Chungbuk | 56.7 | 37.2 | 34.4 | |
| Chungnam | 54.8 | 36.2 | 33.9 | |
| Jeonbuk | 53.9 | 35.8 | 33.6 | |
| Jeonnam | 54.0 | 34.3 | 36.5 | |
| Gyeongbuk | 57.5 | 32.0 | 44.3 | |
| Gyeongnam | 56.1 | 35.0 | 37.6 | |
| Jeju | 60.2 | 39.2 | 34.9 | |
Values are presented as %.
KCDC, Korea Centers for Disease Control and Prevention; COVID-19, coronavirus disease 2019; anti-N, antibodies to nucleocapsid proteins; SARS-COV-2, severe acute respiratory syndrome coronavirus 2.
1 Estimated with age-standardized weights using the population of registered residents in December 2021.
2 The cumulative incidence rate was calculated by using the cumulative number of people confirmed by KCDC to have COVID-19 through July 31, 2022 as the numerator and the registered resident population in December 2021 as the denominator.
- 1. World Health Organization. Rolling updates on coronavirus disease (COVID-19) updated 31 July 2020. [cited 2022 Dec 16]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen.
- 2. CoronaBoard. COVID-19 dashboard. 2022 Jul 31 [cited 2022 Dec 21]. Available from: https://coronaboard.kr/ (Korean).
- 3. Ashcroft P, Lehtinen S, Bonhoeffer S. Test-trace-isolate-quarantine (TTIQ) intervention strategies after symptomatic COVID-19 case identification. PLoS One 2022;17:e0263597.ArticlePubMedPMC
- 4. Yoon K, Kim J, Peck KR, Kim HS, Lee H, Hwang YS, et al. Seroprevalence of SARS-CoV-2 antibodies during the third wave of COVID-19 in the Seoul metropolitan area of Korea. Epidemiol Health 2022;44:e2022085.ArticlePubMedPMCPDF
- 5. Yanes-Lane M, Winters N, Fregonese F, Bastos M, Perlman-Arrow S, Campbell JR, et al. Proportion of asymptomatic infection among COVID-19 positive persons and their transmission potential: a systematic review and meta-analysis. PLoS One 2020;15:e0241536.ArticlePubMedPMC
- 6. Zou L, Ruan F, Huang M, Liang L, Huang H, Hong Z, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med 2020;382:1177-1179.ArticlePubMedPMC
- 7. Lee S, Kim T, Lee E, Lee C, Kim H, Rhee H, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 infection in a community treatment center in the Republic of Korea. JAMA Intern Med 2020;180:1447-1452.ArticlePubMed
- 8. Gao Z, Xu Y, Sun C, Wang X, Guo Y, Qiu S, et al. A systematic review of asymptomatic infections with COVID-19. J Microbiol Immunol Infect 2021;54:12-16.ArticlePubMed
- 9. World Health Organization. A coordinated global research roadmap: 2019 novel coronavirus. 2020 Mar 12 [cited 2022 Dec 22]. Available from: https://www.who.int/publications/m/item/a-coordinated-global-research-roadmap.
- 10. Centers for Disease Control and Prevention. Interim guidelines for COVID-19 antibody testing. 2022 Dec 16 [cited 2022 Dec 27]. Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/testing/antibody-tests-guidelines.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Flab%2Fresources%2Fantibody-tests-guidelines.html.
- 11. Bobrovitz N, Arora RK, Cao C, Boucher E, Liu M, Donnici C, et al. Global seroprevalence of SARS-CoV-2 antibodies: a systematic review and meta-analysis. PLoS One 2021;16:e0252617.ArticlePubMedPMC
- 12. Nah EH, Cho S, Park H, Hwang I, Cho HI. Nationwide seroprevalence of antibodies to SARS-CoV-2 in asymptomatic population in South Korea: a cross-sectional study. BMJ Open 2021;11:e049837.ArticlePubMed
- 13. SeroTracker. Key insights. 2022 Nov [cited 2022 Dec 21]. Available from: https://serotracker.com/en/Explore.Article
- 14. Bergeri I, Whelan MG, Ware H, Subissi L, Nardone A, Lewis HC, et al. Global SARS-CoV-2 seroprevalence from January 2020 to April 2022: a systematic review and meta-analysis of standardized population-based studies. PLoS Med 2022;19:e1004107.ArticlePubMedPMC
- 15. Elliott P, Ward H, Riley S. Population monitoring of SARS-CoV-2 infections via random sampling during the COVID-19 pandemic. Am J Public Health 2023;113:514-516.ArticlePubMedPMC
- 16. Australian COVID-19 Serosurveillance Network. Seroprevalence of SARS-CoV-2-specific antibodies among Australian blood donors: round 3 update. 2022 Nov 3 [cited 2022 Dec 21]. Available from: https://www.kirby.unsw.edu.au/sites/default/files/documents/COVID19-Blood-Donor-Report-Round3-Aug-Sep-2022%5B1%5D.pdf.
- 17. Clarke KE, Jones JM, Deng Y, Nycz E, Lee A, Iachan R, et al. Seroprevalence of infection-induced SARS-CoV-2 antibodies - United States, September 2021-February 2022. MMWR Morb Mortal Wkly Rep 2022;71:606-608.ArticlePubMedPMC
- 18. Akinbami LJ, Kruszon-Moran D, Wang CY, Storandt RJ, Clark J, Riddles MK, et al. SARS-CoV-2 serology and self-reported infection among adults - National Health and Nutrition Examination Survey, United States, August 2021-May 2022. MMWR Morb Mortal Wkly Rep 2022;71:1522-1525.ArticlePubMedPMC
- 19. Song SK, Lee DH, Nam JH, Kim KT, Do JS, Kang DW, et al. IgG seroprevalence of COVID-19 among individuals without a history of the coronavirus disease infection in Daegu, Korea. J Korean Med Sci 2020;35:e269.ArticlePubMedPMCPDF
- 20. Noh JY, Seo YB, Yoon JG, Seong H, Hyun H, Lee J, et al. Seroprevalence of anti-SARS-CoV-2 antibodies among outpatients in southwestern Seoul, Korea. J Korean Med Sci 2020;35:e311.ArticlePubMedPMCPDF
- 21. Kang YW, Ko YS, Kim YJ, Sung KM, Kim HJ, Choi HY, et al. Korea Community Health Survey data profiles. Osong Public Health Res Perspect 2015;6:211-217.ArticlePubMedPMC
- 22. Kim YT, Choi BY, Lee KO, Kim H, Chun JH, Kim SY, et al. Overview of Korean Community Health Survey. J Korean Med Assoc 2012;55:74-83 (Korean).Article
- 23. Kweon S, Kim Y, Jang MJ, Kim Y, Kim K, Choi S, et al. Data resource profile: the Korea National Health and Nutrition Examination Survey (KNHANES). Int J Epidemiol 2014;43:69-77.ArticlePubMedPMC
- 24. Korea Disease Control and Prevention Agency. Korea community health at a glance 2020: Korea Community Health Survey (KCHS). Cheongju: Korea Disease Control and Prevention Agency; 2021. (Korean).
- 25. Muench P, Jochum S, Wenderoth V, Ofenloch-Haehnle B, Hombach M, Strobl M, et al. Development and validation of the Elecsys Anti-SARS-CoV-2 immunoassay as a highly specific tool for determining past exposure to SARS-CoV-2. J Clin Microbiol 2020;58:e01694-e016920.ArticlePubMedPMC
- 26. Gralinski LE, Menachery VD. Return of the coronavirus: 2019-nCoV. Viruses 2020;12:135.ArticlePubMedPMC
- 27. Lee N, Jeong S, Lee SK, Cho EJ, Hyun J, Park MJ, et al. Quantitative analysis of anti-N and anti-S antibody titers of SARS-CoV-2 infection after the third dose of COVID-19 vaccination. Vaccines (Basel) 2022;10:1143.ArticlePubMedPMC
- 28. Rashidzadeh H, Danafar H, Rahimi H, Mozafari F, Salehiabar M, Rahmati MA, et al. Nanotechnology against the novel coronavirus (severe acute respiratory syndrome coronavirus 2): diagnosis, treatment, therapy and future perspectives. Nanomedicine (Lond) 2021;16:497-516.ArticlePubMed
- 29. Korean Law Information Center. Guideline of sampling design and management; 2016 [cited 2022 Dec 20]. Available from: https://www.law.go.kr/LSW/admRulLsInfoP.do?admRulSeq=2100000042881 (Korean).
- 30. Korea Disease Control and Prevention Agency. COVID-19 for healthcare provider. [cited 2022 Dec 20]. Available from: https://ncv.kdca.go.kr/hcp/page.do?mid=0202 (Korean).
- 31. Korea Disease Control and Prevention Agency. COVID-19 vaccination notice: 5-11 years; 2022 [cited 2022 Dec 20]. Available from: https://ncv.kdca.go.kr/menu.es?mid=a10124030000 (Korean).
- 32. Korea Disease Control and Prevention Agency. Urging participation in COVID-19 vaccination for severe illness and mortality prevention in pediatric and adolescent populations. 2022 Aug 18 [cited 2022 Dec 20]. Available from: https://ncov.kdca.go.kr/tcmBoardView.do?brdId=3&brdGubun=31&dataGubun=&ncvContSeq=6826&contSeq=6826&board_id =312&gubun=BDJ (Korean).
- 33. UK Health Security Agency. COVID-19 vaccine surveillance report week 35: 1 September 2022. [cited 2022 Dec 20]. Available from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1101870/vaccine-surveillance-report-week-35.pdf.
- 34. Inbaraj LR, George CE, Chandrasingh S. Seroprevalence of COVID-19 infection in a rural district of South India: a population-based seroepidemiological study. PLoS One 2021;16:e0249247.ArticlePubMedPMC
- 35. Mohammed I, Nauman A, Paul P, Ganesan S, Chen KH, Jalil SM, et al. The efficacy and effectiveness of the COVID-19 vaccines in reducing infection, severity, hospitalization, and mortality: a systematic review. Hum Vaccin Immunother 2022;18:2027160.ArticlePubMedPMC
- 36. Korea Disease Control and Prevention Agency. Urging COVID-19 vaccination for the prevention of severe illness and mortality, including older adults and other eligible individuals. 2022 Jul 28 [cited 2022 Dec 20]. Available from: https://ncov.kdca.go.kr/tcmBoardView.do?brdId=3&brdGubun=31&dataGubun=&ncvContSeq=6784&contSeq=6784&board_id=312&gubun=BDJ (Korean).
- 37. Korea Disease Control and Prevention Agency. The results of COVID-19 antibody prevalence surveys indicate that the natural infection rate is higher than the cumulative incidence rate during the same period. 2022 Jun 14 [cited 2022 Dec 20]. Available from: https://www.kdca.go.kr/board/board.es?mid=a20501010000&bid=0015&act=view&list_no=719832# (Korean).
- 38. Shakiba M, Nazemipour M, Heidarzadeh A, Mansournia MA. Prevalence of asymptomatic COVID-19 infection using a seroepidemiological survey. Epidemiol Infect 2020;148:e300.ArticlePubMed
- 39. Byambasuren O, Cardona M, Bell K, Clark J, McLaws ML, Glasziou P. Estimating the extent of asymptomatic COVID-19 and its potential for community transmission: systematic review and meta-analysis. J Assoc Med Microbiol Infect Dis Can 2020;5:223-234.ArticlePubMedPMC
- 40. Nishiura H, Kobayashi T, Miyama T, Suzuki A, Jung SM, Hayashi K, et al. Estimation of the asymptomatic ratio of novel coronavirus infections (COVID-19). Int J Infect Dis 2020;94:154-155.ArticlePubMedPMC
- 41. Abedi V, Olulana O, Avula V, Chaudhary D, Khan A, Shahjouei S, et al. Racial, economic, and health inequality and COVID-19 infection in the United States. J Racial Ethn Health Disparities 2021;8:732-742.ArticlePubMedPDF
- 42. Garnier R, Benetka JR, Kraemer J, Bansal S. Socioeconomic disparities in social distancing during the COVID-19 pandemic in the United States: observational study. J Med Internet Res 2021;23:e24591.ArticlePubMedPMC
- 43. Krutikov M, Palmer T, Tut G, Fuller C, Azmi B, Giddings R, et al. Prevalence and duration of detectable SARS-CoV-2 nucleocapsid antibodies in staff and residents of long-term care facilities over the first year of the pandemic (VIVALDI study): prospective cohort study in England. Lancet Healthy Longev 2022;3:e13-e21.ArticlePubMedPMC
- 44. Nakagama Y, Komase Y, Kaku N, Nitahara Y, Tshibangu-Kabamba E, Tominaga T, et al. Detecting waning serological response with commercial immunoassays: 18-month longitudinal follow-up of anti-SARS-CoV-2 nucleocapsid antibodies. Microbiol Spectr 2022;10:e0098622.ArticlePubMedPDF
- 45. Schallier A, De Baets S, De Bruyne D, Dauwe K, Herpol M, Couck P. Assay dependence of long-term kinetics of SARS-CoV-2 antibodies. Diagn Microbiol Infect Dis 2021;100:115403.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Infection-mediated immune response in SARS-CoV-2 breakthrough infection and implications for next-generation COVID-19 vaccine development
Sho Miyamoto, Tadaki Suzuki
Vaccine.2024; 42(6): 1401. CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite