Articles
- Page Path
- HOME > Epidemiol Health > Volume 39; 2017 > Article
-
Original Article
Cancer mortality-to-incidence ratio as an indicator of cancer management outcomes in Organization for Economic Cooperation and Development countries -
Eunji Choi1
 , Sangeun Lee1
, Sangeun Lee1 , Bui Cam Nhung1
, Bui Cam Nhung1 , Mina Suh2
, Mina Suh2 , Boyoung Park1,2
, Boyoung Park1,2 , Jae Kwan Jun1,2
, Jae Kwan Jun1,2 , Kui Son Choi1,2
, Kui Son Choi1,2
-
Epidemiol Health 2017;39:e2017006.
DOI: https://doi.org/10.4178/epih.e2017006
Published online: February 5, 2017
1Graduate School of Cancer Science and Policy, National Cancer Center, Goyang, Korea
2National Cancer Control Institute, National Cancer Center, Goyang, Korea
- Correspondence: Kui Son Choi Graduate School of Cancer Science and Policy, National Cancer Center, 323 Ilsan-ro, Ilsandong-gu, Goyang 10408, Korea E-mail: kschoi@ncc.re.kr
• Received: January 5, 2017 • Accepted: February 5, 2017
©2017, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES
- Assessing long-term success and efficiency is an essential part of evaluating cancer control programs. The mortality-to-incidence ratio (MIR) can serve as an insightful indicator of cancer management outcomes for individual nations. By calculating MIRs for the top five cancers in Organization for Economic Cooperation and Development (OECD) countries, the current study attempted to characterize the outcomes of national cancer management policies according to the health system ranking of each country.
-
METHODS
- The MIRs for the five most burdensome cancers globally (lung, colorectal, prostate, stomach, and breast) were calculated for all 34 OECD countries using 2012 GLOBOCAN incidence and mortality statistics. Health system rankings reported by the World Health Organization in 2000 were updated with relevant information when possible. A linear regression model was created, using MIRs as the dependent variable and health system rankings as the independent variable.
-
RESULTS
- The linear relationships between MIRs and health system rankings for the five cancers were significant, with coefficients of determination ranging from 49 to 75% when outliers were excluded. A clear outlier, Korea reported lower-than-predicted MIRs for stomach and colorectal cancer, reflecting its strong national cancer control policies, especially cancer screening.
-
CONCLUSIONS
- The MIR was found to be a practical measure for evaluating the long-term success of cancer surveillance and the efficacy of cancer control programs, especially cancer screening. Extending the use of MIRs to evaluate other cancers may also prove useful.
- Cancer is a leading cause of death in both more and less economically developed countries. In 2012, there were 14.1 million new cancer cases and 8.2 million cancer deaths worldwide; 57% (8 million) of new cancer cases and 65% (5.3 million) of cancer deaths occurred in less developed regions [1]. Due to population growth and aging, the global cancer burden is expected to grow. The five most common cancers (lung, breast, colorectal, prostate, and stomach) in both sexes account for nearly half of all cancer cases. Lung and breast cancer are the most frequently diagnosed cancers, and are the leading causes of cancer death in men and women, respectively, both overall and in less developed countries [2]. In general, cancer incidence rates are higher in more developed regions with longer life expectancies. In fact, the incidence rates for all cancers combined are twice as high for more developed countries than for less developed countries. However, mortality rates for all cancers are only 8 to 15% higher in more developed countries [2]. This disparity primarily reflects differences in the distribution of cancer cases, which is affected by risk factors, detection practices, and/or the availability of treatment.
- A substantial portion of cancer cases and deaths could be prevented by broadly applying effective prevention measures, such as tobacco control, vaccination, and the use of early detection tests. Thus, the implementation of cancer control programs has been recommended as a means to effectively reduce cancer incidence and mortality, and national cancer control programs have been developed in several countries [3]. Nonetheless, assessing the long-term success and efficiency of these programs is essential. The mortality-to-incidence ratio (MIR) provides an alternative means to assess the burden of a disease by presenting mortality after accounting for incidence. In prior studies, the MIR was found to be a simple and insightful measure of the efficacy of cancer control programs [4,5]. The ratio identifies whether a country has a higher or lower mortality for a condition, normalized to its incidence. To determine the causes of differences in mortality and incidence, other information should be gathered. Previously, the MIR statistic has been used to demonstrate racial disparities in cancers [6], as well as to examine relationships between health care systems and cancer outcomes in the US [7] and worldwide [8]. Recently, Sunkara & Hébert [8] demonstrated a strong association between MIRs for colorectal cancer and the quality of health care systems. They suggested that the MIR could be useful as an indicator for identifying disparities in cancer screening and treatment internationally.
- Therefore, in this study, we calculated MIRs for the five most common cancers in the 34 Organization for Economic Cooperation and Development (OECD) member countries in an attempt to evaluate the outcomes of national cancer management policies according to the performance of each country’s health system. Only OECD member countries were chosen because of their high-quality health care-related data. In particular, this study aimed to assess the outcomes of cancer control programs in Korea, as reflected by the MIR, in comparison to MIR values and health care system rankings across OECD countries. Additionally, we attempted to identify factors that could potentially explain outliers, in which MIRs were not well predicted by regression models.
INTRODUCTION
- Mortality and incidence rate data
- Mortality and incidence rate data were derived from the GLOBOCAN 2012 database for all 34 OECD countries [1]. The GLOBOCAN database provides contemporary estimates of the incidence, mortality, and prevalence of major types of cancer at the national level for 184 countries throughout the world. We collected the age-standardized rates per 100,000 population per year for lung, colorectal, prostate, stomach, and breast cancer, and calculated the MIRs by dividing the mortality rate by the incidence rate. When using the GLOBOCAN data, it is recommended to report the scope of the data sources and methods. In that database, the quality of the data on the incidence rate is graded from A (high quality) to G (no data), depending on the availability of incidence data. For grade G countries, GLOBOCAN contains estimated incidence rates using those of neighboring countries or registries in the same area [1]. Similarly, for mortality rates, data are scored from 1 (high quality, complete registration) to 6 (no data). In our data set of OECD countries, the mean grade of incidence rate data was grade B; only data for Greece and Hungary were given a grade of G. The mean score for the available mortality data was 1.79 (from 1 to 6), with Mexico reporting the highest score of five. Despite the poor quality of incidence or mortality data from Greece, Hungary, and Mexico, the methods used to estimate cancer incidence and mortality are well established and reported in the GLOBOCAN database. We therefore included all these countries in the analysis, in order to compare MIRs for the five most common cancers across the 34 OECD member countries. Moreover, the results were very similar, whether or not we included data from countries with poor-quality data (Mexico, Hungary, and Greece) in the analysis.
- Health system rankings
- As an indicator of the quality of health systems, we adopted the health system rankings presented by the World Health Organization (WHO) in the year 2000 for 191 countries [9]. The health system ranking reflects five composite measures: overall health, health care financing, health inequality, health responsiveness, and distribution of health care services. Data for the five composite measures were derived from estimates for each country in 1997. Although the rankings have not been updated due to criticisms about their efficacy [10], we valued the methodological framework and the thoroughness of the data based on which the indicators were developed [11]. We decided to use the rankings after updating the composite measures when possible. Among the five composite measures, it was only possible to update overall health and health inequality. As overall health in the initial health ranking report was represented by health-adjusted life expectancy (HALE), we updated the measure with 2012 HALE data [12]. Health inequality was derived from calculations on child mortality [11]; therefore, we adopted 2011 OECD health data on child mortality. The other three measures (health care financing, health responsiveness, and distribution of health care services) were not able to be updated due to inconsistencies in the data available for all OECD countries. Our updated version of the health system rankings reflected rankings similar to those originally reported, except for Austria, Portugal, Greece, Switzerland, Canada, Australia, New Zealand, and Korea.
- Statistical analysis
- A simple linear regression model was generated by taking the MIR as a dependent variable and the updated health system rankings as the independent variable. For the analysis, not the exact values of each health system ranking for countries, but the ranking number itself was used for the analysis, as previous studies have confirmed the presence of a linear association between the MIR and the health system ranking itself [8]. The formula for this analysis was as follows: predicted MIR= health system ranking× beta+alpha. Divergent points were then identified. Divergent points were defined as countries for which the residuals between their actual MIR and their predicted MIR determined by the regression model were greater or less than 0.07. After defining divergent points, we performed an additional simple linear regression analysis excluding divergent countries. Statistical analyses were performed using Stata version 13 (StataCorp., College Station, TX, USA), taking p-values < 0.05 to indicate statistical significance.
- Ethical issues
- This study was exempted from the institutional ethics review board, because it was not human-subjects research and analyzed existing data.
MATERIALS AND METHODS
- Figure 1 (A-E) depicts scatter plots with predicted lines for lung, colorectal, prostate, stomach, and breast cancer. For all scatter plots, we detected significant linear relationships between the MIR and the health system rankings, with coefficients of determination ranging from 32 to 55%. These results demonstrated a positive association between lower health care system rankings (1-unit changes) and higher MIRs.
- For lung cancer (Figure 1A), with every 1-unit change in health system ranking, there was a 0.004 increment rise in the MIR. Eight countries were identified as divergent points in the lung cancer model: the Slovak Republic, Czech Republic, US, and Australia demonstrated lower MIRs than predicted, whereas Sweden, Italy, Chile, and Estonia showed higher MIRs. Figure 1B presents a 0.007 incremental change in MIR for colorectal cancer with a 1-unit change in the health system ranking. Divergent points for this model included Denmark, Iceland, Korea, and Belgium, all of which had lower MIRs than predicted, and Spain, Poland, Japan, Turkey, Chile, and Greece, which had higher MIRs. In the prostate cancer model, a 1-unit change in health system ranking generated an increase in MIR of 0.007 units (Figure 1C). The US, Czech Republic, Ireland, Estonia, Finland, Israel, and Portugal had lower MIRs than predicted, while Chile, Japan, Mexico, Greece, and Turkey had higher MIRs than predicted in this model. For stomach cancer (Figure 1D), every 1-unit change in health system ranking led to an increase in MIR of 0.008 units. Its divergent points corresponding to a lower-than-predicted MIR were Korea, Denmark, the United States, Czech Republic, Luxembourg, Japan, Slovak Republic, and Estonia; higher-than-predicted MIRs were found for Spain, Turkey, Poland, Switzerland, Greece, Italy, Sweden, and Chile. Finally, for breast cancer (Figure 1E), a 1-unit change in health system ranking increased the MIR by 0.004 units. Among the divergent points for breast cancer, the Czech Republic exhibited a lower MIR, while Turkey, Chile, and Greece showed higher MIRs than predicted. Appendices 1-5 present the complete data on the updated health system rankings, mortality rates, incidence rates, actual MIRs, predicted MIRs, and residuals, alphabetically sorted by country name.
- To eliminate the effect of divergent points, we excluded countries with residuals between their actual MIR and their predicted MIR that were greater or less than 0.07. Table 1 lists the coefficients of determination for the original model and the additional model devised after eliminating the divergent points. The R2 value for lung cancer in the original model was 0.32 (meaning that 32% of the total variability in MIR for lung cancer was explained by the model), and it increased to 0.49 after removing outliers. The R2 value for colorectal cancer increased from 0.55 to 0.68; the R2 value for prostate cancer increased from 0.41 to 0.75; the R2 value for stomach cancer increased from 0.40 to 0.73; and the R2 value for breast cancer increased from 0.51 to 0.55.
RESULTS
- In the present study, we demonstrated a significant positive linear relationship between the MIR and the updated health care system rankings. After removing divergent points, we detected substantial increases in the coefficients of determination for each cancer model, up to 0.75 for prostate cancer, meaning that 75% of the total variability in the MIR across countries was explained by the updated health care system rankings. In the lung cancer model, however, the coefficient of determination remained only at 0.49. Despite improvements in cancer treatment, the overall survival rate for lung cancer remains around 20% [13]. Additionally, although lung cancer screening with low-dose computed tomography is now recommended in several guidelines, researchers have yet to alleviate concerns about the sensitivity of the test [14]. Therefore, differences in the MIR for lung cancer among OECD countries might not be clearly explained by differences in health systems.
- In the models for stomach and colorectal cancer, Korea was a clear divergent point, with MIRs that were much lower than predicted. While the average MIR among all OECD countries was 0.63 for stomach cancer, Korea reported an MIR of 0.31. In the colorectal cancer model, Korea’s MIR was 0.23, compared to the average MIR of 0.38. We suspect that the low MIRs for Korea reflect the nation’s strong national cancer control policies. In Korea, cancer is responsible for one in every four deaths [15]. In an effort to reduce the increasing cancer burden, the Korean government has supported cancer screening via the National Cancer Screening Program (NCSP) for the Korean population since 2002. Via the NCSP, medical aid enrollees and the lower 50% of income bracket among the National Health Insurance (NHI) beneficiaries are eligible for free-of-charge screening for stomach, breast, cervix, liver, and colorectal cancer. The more affluent 50% of NHI beneficiaries are eligible for screening with a co-payment of 10%. For detecting stomach cancer, eligible participants over the age of 40 years are invited biennially to undergo screening via upper endoscopy or upper gastrointestinal series. The total screening rate for stomach cancer was 73.6% in 2013 [16]. For colorectal cancer, individuals over 50 years of age are annually invited to undergo an initial mass screening with a fecal occult blood test, and a further examination with colonoscopy or double-contrast barium enema is provided for those with positive results. The screening rate for colorectal cancer was 55.6% in 2013 [16]. According to our results, we suggest that the nationwide cancer screening program in Korea appears to be associated with an MIR lower than that predicted by the regression model.
- Similar implications are also applicable for other divergent points in the regression models. In Japan, stomach cancer is a serious burden, accounting for 14.2% of all cancer deaths [17]. To reduce this burden, Japan has also conducted stomach cancer screening with photofluorography as part of a national program. Under the national health policy for the prevention of chronic diseases, stomach cancer screening has been promoted by providing financial support for cancer screenings. In the present study, Japan showed a lower-than-predicted MIR for stomach cancer of 0.41. In contrast, the higher-than-predicted MIRs among divergent nations may stem from a lack of appropriate cancer control programs. For example, Chile, which also reports one of the highest incidence rates of stomach cancer, lacks screening guidelines for stomach cancer, though it has implemented a national integrated non-communicable disease policy and action plans [8]. Likewise, for colorectal cancer, Denmark, Iceland, and Belgium showed lower-than-predicted MIRs, and all have formal screening recommendations for colorectal cancer in place [18]. Meanwhile, countries with higher-than-predicted MIRs were less likely to have formal screening recommendations or tended to have lower screening rates for colorectal cancer [8].
- Unexpectedly, Korea was not classified as a divergent nation in the breast cancer model, though it has provided biennial breast cancer screening with a mammography for all women over 40 years under the NCSP. For breast cancer, the majority of OECD countries conduct mammography screenings, with relatively high screening rates. In addition, the treatment of breast cancer has improved greatly with the introduction of multidisciplinary breast cancer care units, reducing the benefits from mammography screening. Still, Korea reported a lower actual MIR of 0.11 for breast cancer than its predicted MIR of 0.15, which is also lower than the average MIR across OECD countries of 0.20.
- The NCSP in Korea does not provide nationwide screening for lung and prostate cancer. Nevertheless, the nation still recorded an actual MIR for lung cancer of 0.74, lower than its predicted value of 0.76 and lower than the average value for all OECD countries of 0.80. This might be explained by Korea’s comparatively high 5-year survival rates for lung cancer. Korea had a 5-year survival rate for lung cancer of 20.7%, while the 5-year survival rates were 16.6% in the US, 17% in Canada, and 29.7% in Japan [15,19,20]. For prostate cancer, Korea reported a higher actual MIR of 0.15 than the predicted value of 0.11. In comparison, the actual MIR for prostate cancer in the US was 0.10, the lowest among all OECD countries. In the US, prostate cancer is the most common cancer and the second leading cause of death among men, according to the National Cancer Institute statistics [21]. To the reduce cancer burden, prostate cancer screening is recommended by the American Cancer Society with informed consent, although the US Preventive Services Task Force has warned against prostate cancer screening because its harms may outweigh its benefits. Nevertheless, the guidelines and screening programs for prostate cancer proposed by the American Cancer Society seem to have helped effectively control prostate cancer, as reflected by its low MIR [22].
- Our study has several limitations that warrant consideration. First, our data focused wholly on OECD countries, which generally have more sound health infrastructure. This limits the generalizability of our results to low-income and middle-income countries lacking the needed infrastructure. Second, there were inconsistencies in the data sources and methods for determining cancer mortality and incidence rates from GLOBOCAN, as described in the Methods section. Nonetheless, our findings were consistent regardless of whether we included data from countries with poor-quality data. Furthermore, updating the data for the WHO 2000 health system rankings was not fully achieved due to a lack of available data. Thus, our rankings may not exactly reflect the most recent performance of each nation’s health care system.
- In this study, we found that lower MIRs reflected the implementation of effective cancer control programs, including cancer screening. In contrast, countries with higher-than-predicted MIRs often lacked proper health policies or recommendations for cancer control. For Korea, among the five cancers analyzed in this study, stomach and colorectal cancer had markedly low MIRs, indicating effective cancer control, mainly as a result of screening programs offered via the NCSP. Despite finding the MIR to be an efficient and useful indicator of cancer control outcomes, studies on mortality rate reductions are required to confirm the effectiveness of cancer control. Notwithstanding, we favor extending the use of the MIR for other cancers to assess the long-term success of cancer screening programs.
DISCUSSION
ACKNOWLEDGEMENTS
Figure 1.Mortality-to-incidence ratio (MIR) for the five most common cancers (A: lung, B: colorectal, C: prostate, D: stomach, and E: breast) vs. health system ranking for all 34 Organization for Economic Cooperation and Development countries.
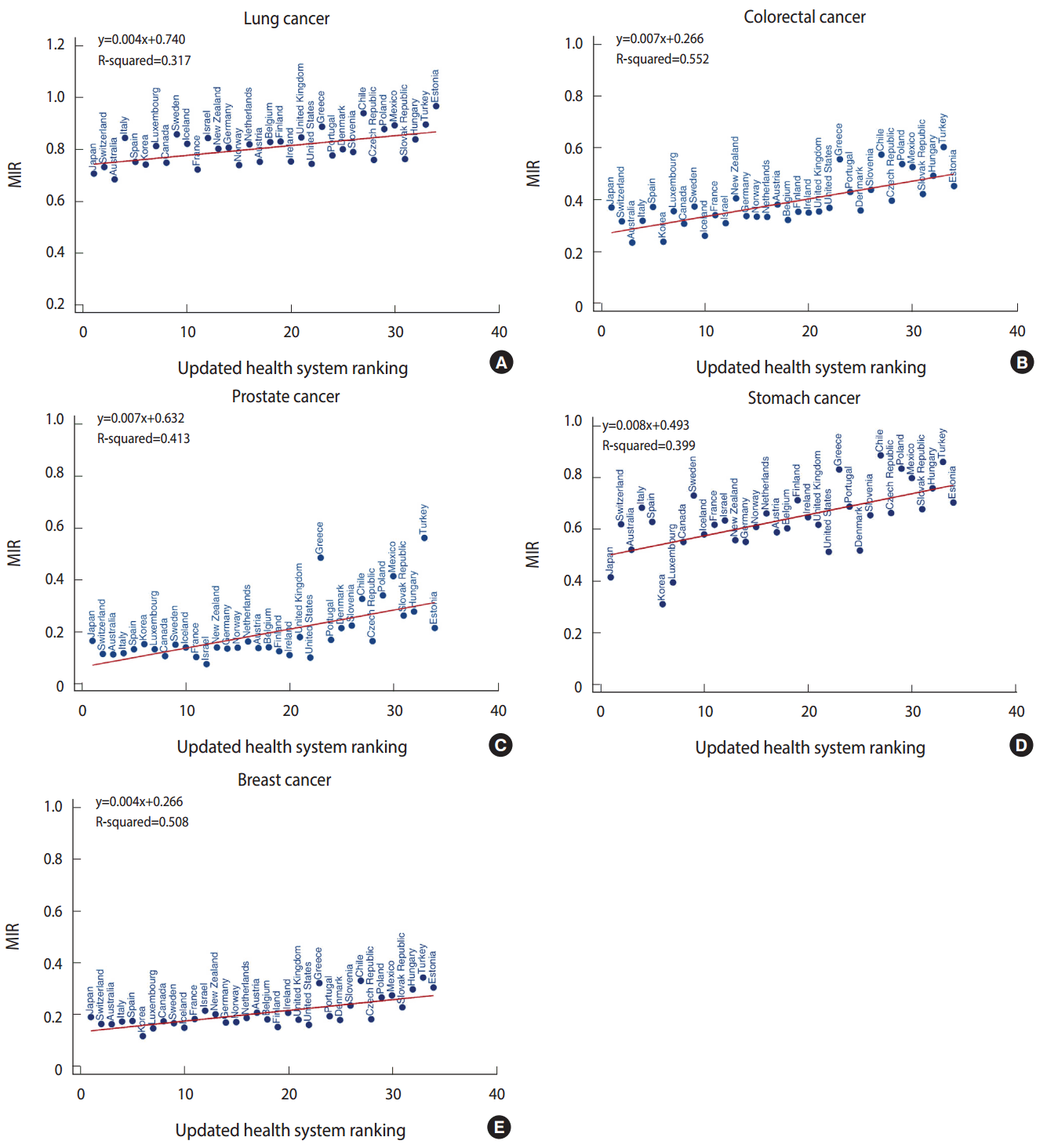

Table 1.Coefficients of determination before and after removing outliers
| Cancer | R2 in original models | R2 without outliers |
|---|---|---|
| Lung | 0.32 | 0.49 |
| Colorectal | 0.55 | 0.68 |
| Prostate | 0.41 | 0.75 |
| Stomach | 0.40 | 0.73 |
| Breast | 0.51 | 0.55 |
- 1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359-E386.ArticlePubMed
- 2. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108.ArticlePubMed
- 3. World Health Organization. Cancer control: knowledge into action. WHO guide for effective programmes. [cited 2017 Feb 24]. Available from: http://www.who.int/cancer/modules/en/.
- 4. Asadzadeh Vostakolaei F, Karim-Kos HE, Janssen-Heijnen ML, Visser O, Verbeek AL, Kiemeney LA. The validity of the mortality to incidence ratio as a proxy for site-specific cancer survival. Eur J Public Health 2011;21:573-577.ArticlePubMedPDF
- 5. Parkin DM, Bray F. Evaluation of data quality in the cancer registry: principles and methods Part II. Completeness. Eur J Cancer 2009;45:756-764.ArticlePubMed
- 6. Hébert JR, Daguise VG, Hurley DM, Wilkerson RC, Mosley CM, Adams SA, et al. Mapping cancer mortality-to-incidence ratios to illustrate racial and sex disparities in a high-risk population. Cancer 2009;115:2539-2552.ArticlePubMedPMC
- 7. Adams SA, Choi SK, Khang L, A Campbell D, Friedman DB, Eberth JM, et al. Decreased cancer mortality-to-incidence ratios with increased accessibility of federally qualified health centers. J Community Health 2015;40:633-641.ArticlePubMedPMC
- 8. Sunkara V, Hébert JR. The colorectal cancer mortality-to-incidence ratio as an indicator of global cancer screening and care. Cancer 2015;121:1563-1569.ArticlePubMedPMC
- 9. Ajay Tandon A, Murray CJ, Lauer JA, Evans DB. Measuring overall health system performance for 191 countries. [cited 2017 Feb 25]. Available from: http://www.who.int/healthinfo/paper30.pdf.
- 10. Jamison DT, Sandbu ME. Global health. WHO ranking of health system performance. Science 2001;293:1595-1596.ArticlePubMed
- 11. Murray CJ, Frenk J. A WHO framework for health system performance assessment. [cited 2017 Feb 24]. Available from: http://www.who.int/healthinfo/paper06.pdf.
- 12. World Health Organization. WHO methods for life expectancy and healthy life expectancy; 2014 [cited 2017 Feb 24]. Available from: http://www.who.int/healthinfo/statistics/LT_method_1990_2012.pdf.
- 13. National Cancer Institute. Previous version: SEER cancer statistics review, 1975-2010; 2013 [cited 2017 Feb 24]. Available from: https://seer.cancer.gov/archive/csr/1975_2010/.
- 14. Kanodra NM, Silvestri GA, Tanner NT. Screening and early detection efforts in lung cancer. Cancer 2015;121:1347-1356.ArticlePubMed
- 15. Jung KW, Won YJ, Kong HJ, Oh CM, Cho H, Lee DH, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2012. Cancer Res Treat 2015;47:127-141.ArticlePubMedPMCPDF
- 16. Suh M, Choi KS, Lee YY, Jun JK. Trends in cancer screening rates among Korean men and women: results from the Korean National Cancer Screening Survey, 2004-2012. Cancer Res Treat 2013;45:86-94.ArticlePubMedPMCPDF
- 17. Katanoda K, Matsuda T, Matsuda A, Shibata A, Nishino Y, Fujita M, et al. An updated report of the trends in cancer incidence and mortality in Japan. Jpn J Clin Oncol 2013;43:492-507.ArticlePubMedPDF
- 18. Altobelli E, Lattanzi A, Paduano R, Varassi G, di Orio F. Colorectal cancer prevention in Europe: burden of disease and status of screening programs. Prev Med 2014;62:132-141.ArticlePubMed
- 19. Kachuri L, De P, Ellison LF, Semenciw R; Advisory Committee on Canadian Cancer Statistics. Cancer incidence, mortality and survival trends in Canada, 1970-2007. Chronic Dis Inj Can 2013;33:69-80.ArticlePubMed
- 20. Matsuda T, Marugame T, Kamo K, Katanoda K, Ajiki W, Sobue T, et al. Cancer incidence and incidence rates in Japan in 2005: based on data from 12 population-based cancer registries in the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol 2011;41:139-147.ArticlePubMedPDF
- 21. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin 2008;58:71-96.ArticlePubMed
- 22. Mettlin C, Lee F, Drago J, Murphy GP. The American Cancer Society National Prostate Cancer Detection Project. Findings on the detection of early prostate cancer in 2425 men. Cancer 1991;67:2949-2958.ArticlePubMed
REFERENCES
Appendix
Figure & Data
References
Citations
Citations to this article as recorded by 

- Global burden of 34 cancers among women in 2020 and projections to 2040: Population‐based data from 185 countries/territories
Habtamu Mellie Bizuayehu, Abel F. Dadi, Tahir A. Hassen, Daniel Bekele Ketema, Kedir Y. Ahmed, Zemenu Y. Kassa, Erkihun Amsalu, Getiye Dejenu Kibret, Addisu Alehegn Alemu, Animut Alebel, Jemal E. Shifa, Yibeltal Assefa, Gizachew A. Tessema, Peter Sarich,
International Journal of Cancer.2024; 154(8): 1377. CrossRef - Gastric cancer screening in Western countries: A call to action
Fabio Farinati, Filippo Pelizzaro
Digestive and Liver Disease.2024;[Epub] CrossRef - Development, acceptability and usability of culturally appropriate survivor narrative videos for breast cancer treatment in Botswana: a pilot study
Yehoda M Martei, Lebogang Mokokwe, Ngwao Ngwako, Keaobaka Kebuang, Dipho I Setlhako, Goitsemang Gabaatlhole, Bontswanetse Baaitse, Tumisang Segadimo, Lawrence N Shulman, Frances Barg, Babe E Gaolebale
BMJ Open.2024; 14(1): e073867. CrossRef - Disparities in mortality risk after diagnosis of hematological malignancies in 185 countries: A global data analysis
Jing Yang, Xin Liu, Qiu-Zi Zhong, Yong Yang, Tao Wu, Si-Ye Chen, Bo Chen, Yong-Wen Song, Hui Fang, Shu-Lian Wang, Yue-Ping Liu, Jing Jin, Ning Li, Ning-Ning Lu, Hao Jing, Yuan Tang, Fan Chen, Xi-Mei Zhang, Wenwen Zhang, Yirui Zhai, Shu-Nan Qi, Ye-Xiong Li
Cancer Letters.2024; 595: 216793. CrossRef - Utility of auto fluorescence-guided biopsy in suspected lung cancer patients with bronchial mucosal lesions
Gaozhe Wang, Yan Jin, Kunlong Xiong, Xiaoyan Jin, Linfeng Wang, Chenwei Li
Photodiagnosis and Photodynamic Therapy.2024; 46: 104057. CrossRef - Policy strategies for capacity building and scale up of the workforce for comprehensive cancer care: a systematic review
D. Trapani, S.S. Murthy, N. Hammad, R. Casolino, D.C. Moreira, F. Roitberg, J.-Y. Blay, G. Curigliano, A.M. Ilbawi
ESMO Open.2024; 9(4): 102946. CrossRef - Global, regional, and national burdens of early onset pancreatic cancer in adolescents and adults aged 15–49 years from 1990 to 2019 based on the Global Burden of Disease Study 2019: a cross-sectional study
Zheng Li, Xiaojie Zhang, Chongyuan Sun, Zefeng Li, He Fei, Dongbing Zhao
International Journal of Surgery.2024; 110(4): 1929. CrossRef - Self-Assembly of Peptides as an Alluring Approach toward Cancer Treatment and Imaging
Muhammad Hamza Ghafoor, Ben-Li Song, Lei Zhou, Zeng-Ying Qiao, Hao Wang
ACS Biomaterials Science & Engineering.2024; 10(5): 2841. CrossRef - Global, regional, and national trends of dementia incidence and risk factors, 1990–2019: A Global Burden of Disease study
Abolfazl Avan, Vladimir Hachinski
Alzheimer's & Dementia.2023; 19(4): 1281. CrossRef - Improved Trends in the Mortality-to-Incidence Ratios for Liver Cancer in Countries with High Development Index and Health Expenditures
Chang-Cheng Su, Brian-Shiian Chen, Hsin-Hung Chen, Wen-Wei Sung, Chi-Chih Wang, Ming-Chang Tsai
Healthcare.2023; 11(2): 159. CrossRef - Updated epidemiology of gastrointestinal cancers in East Asia
Junjie Huang, Don Eliseo Lucero-Prisno, Lin Zhang, Wanghong Xu, Sunny H. Wong, Siew C. Ng, Martin C. S. Wong
Nature Reviews Gastroenterology & Hepatology.2023; 20(5): 271. CrossRef - Peptide Self-Assembled Nanocarriers for Cancer Drug Delivery
Vijay Bhooshan Kumar, Busra Ozguney, Anastasia Vlachou, Yu Chen, Ehud Gazit, Phanourios Tamamis
The Journal of Physical Chemistry B.2023; 127(9): 1857. CrossRef - Stigma and Social Determinants of Health Associated With Fidelity to Guideline-Concordant Therapy in Patients With Breast Cancer Living With and Without HIV in Botswana
Yehoda M Martei, Modesty Obasohan, Lebogang Mokokwe, Tlotlo Ralefala, Mosepele Mosepele, Robert Gross, Frances K Barg
The Oncologist.2023; 28(12): e1230. CrossRef - Clinical Guidance on the Monitoring and Management of Trastuzumab Deruxtecan (T-DXd)-Related Adverse Events: Insights from an Asia-Pacific Multidisciplinary Panel
Joanne Wing Yan Chiu, Soo Chin Lee, James Chung-man Ho, Yeon Hee Park, Ta-Chung Chao, Sung-Bae Kim, Elgene Lim, Ching-Hung Lin, Sherene Loi, Su Ying Low, Lynette Li San Teo, Winnie Yeo, Rebecca Dent
Drug Safety.2023; 46(10): 927. CrossRef - Gender differences in leukemia outcomes based on health care expenditures using estimates from the GLOBOCAN 2020
Maedeh Amini, Rajesh Sharma, Chinmay Jani
Archives of Public Health.2023;[Epub] CrossRef - Unfavorable cancer mortality‐to‐incidence ratios in patients with schizophrenia: A nationwide cohort study in Taiwan, 2000–2019
Chu‐Syuan Cheng, Wen‐Yin Chen, Hu‐Ming Chang, Chun‐Hung Pan, Sheng‐Siang Su, Shang‐Ying Tsai, Chiao‐Chicy Chen, Chian‐Jue Kuo
Acta Psychiatrica Scandinavica.2023; 148(4): 347. CrossRef - Mortality‐to‐incidence ratio of early‐onset colorectal cancer in high‐income Asian and Middle Eastern countries: A systemic analysis of the Global Burden of Diseases Study 2019
Majed Ramadan, Rawiah A. Alsiary, Doaa A. Aboalola
Cancer Medicine.2023; 12(21): 20604. CrossRef - Spiritual coping strategies: Relationship with physical, anxiety, and depression symptoms of patients with cancer
Atefeh Ahmadi, Aazam Heidarzadeh, Mahlagha Dehghan, Mohammah Hossein Safarizadeh, Mansooreh Azizzadeh Forouzi
Indian Journal of Cancer.2023; 60(3): 439. CrossRef - Assessing the quality of care for skin malignant melanoma on a global, regional, and national scale: a systematic analysis of the global burden of disease study from 1990 to 2019
Mingjuan Liu, Mengyin Wu, Xintong Liu, Jia Zhou, Yining Lan, Hanlin Zhang, Xinyi zhang, Ling Leng, Heyi Zheng, Jun Li
Archives of Dermatological Research.2023; 315(10): 2893. CrossRef - The fraction of life years lost after diagnosis (FLYLAD): a person-centred measure of cancer burden
David Banham, Jonathan Karnon, Alex Brown, David Roder, John Lynch
Population Health Metrics.2023;[Epub] CrossRef - Global trend analysis of diabetes mellitus incidence, mortality, and mortality-to-incidence ratio from 1990 to 2019
Mohammadreza Balooch Hasankhani, Hossein Mirzaei, Ali Karamoozian
Scientific Reports.2023;[Epub] CrossRef - Burden of AML, 1990-2019: Estimates From the Global Burden of Disease Study
Chinmay T. Jani, Alaaeldin Ahmed, Harpreet Singh, Christian Mouchati, Omar Al Omari, Padmanabh S. Bhatt, Rajesh Sharma, Minaam Farooq, Weitao Liu, Joseph Shalhoub, Dominic Marshall, Justin D. Salciccioli, Jeremy L. Warner, Prudence Lam
JCO Global Oncology.2023;[Epub] CrossRef - Type 1 diabetes mellitus disease burden in high health expenditure countries between 1990 and 2019
Michael Edwards, Aurimas Kudzinskas, Andrew Alazawi, Will Hughes, Richard Goodall, Eleanor Harbinson, Justin Salciccioli, Dominic Marshall, Joseph Shalhoub
Diabetes and Vascular Disease Research.2023;[Epub] CrossRef - Spatial and temporal patterns of prostate cancer burden and their association with Socio‐Demographic Index in Asia, 1990–2019
Li‐Sha Luo, Jun‐Feng Jiang, Hang‐Hang Luan, Hao Zi, Cong Zhu, Bing‐Hui Li, Xian‐Tao Zeng
The Prostate.2022; 82(2): 193. CrossRef - Cancer of unknown primary ‐ incidence, mortality trend, and mortality‐to‐incidence ratio is associated with human development index in Central Serbia, 1999–2018: Evidence from the national cancer registry
Aleksandra Ignjatović, Miodrag Stojanović, Zoran Milošević, Marija Anđelković Apostolović, Tamara Filipović, Nataša Rančić, Roberta Marković, Marija Topalović, Dijana Stojanović, Suzana Otašević
European Journal of Cancer Care.2022;[Epub] CrossRef - Global, regional, and national burden and quality of care index of endocarditis: the global burden of disease study 1990–2019
Sara Momtazmanesh, Sahar Saeedi Moghaddam, Elaheh Malakan Rad, Sina Azadnajafabad, Narges Ebrahimi, Esmaeil Mohammadi, Mahtab Rouhifard, Negar Rezaei, Masoud Masinaei, Nazila Rezaei, Mohammad Keykhaei, Arya Aminorroaya, Azin Ghamari, Bagher Larijani, Fars
European Journal of Preventive Cardiology.2022; 29(8): 1287. CrossRef - Global pattern of trends in incidence, mortality, and mortality-to-incidence ratio rates related to liver cancer, 1990–2019: a longitudinal analysis based on the global burden of disease study
Maedeh Amini, Mehdi Azizmohammad Looha, Elaheh Zarean, Mohamad Amin Pourhoseingholi
BMC Public Health.2022;[Epub] CrossRef - Mapping Cancer in Africa: A Comprehensive and Comparable Characterization of 34 Cancer Types Using Estimates From GLOBOCAN 2020
Rajesh Sharma, Aashima, Mehak Nanda, Claudio Fronterre, Paul Sewagudde, Anna E. Ssentongo, Kelsey Yenney, Nina D. Arhin, John Oh, Forster Amponsah-Manu, Paddy Ssentongo
Frontiers in Public Health.2022;[Epub] CrossRef - Cancer in sub-Saharan Africa in 2020: a review of current estimates of the national burden, data gaps, and future needs
Freddie Bray, D Maxwell Parkin, Freddy Gnangnon, Gontse Tshisimogo, Jean-Felix Peko, Innocent Adoubi, Mathewos Assefa, Lamin Bojang, Baffour Awuah, Moussa Koulibaly, Nathan Buziba, Anne Korir, Charles Dzamalala, Bakarou Kamate, Shyam Manraj, Josefo Ferro,
The Lancet Oncology.2022; 23(6): 719. CrossRef - Burden of type 1 diabetes mellitus in the North Africa and Middle East Region, 1990–2019; findings from the global burden of disease study
Shahnaz Esmaeili, Sahar Saeedi Moghaddam, Nazli Namazi, Fatemeh Bandarian, Zahra Esfahani, Maryam Peimani, Sarvenaz Shahin, Ensieh Nasli-Esfahani, Isa Akbarzadeh, Ali Ghanbari, Negar Rezaei, Nazila Rezaei, Bagher Larijani, Farshad Farzadfar
Diabetes Research and Clinical Practice.2022; 188: 109912. CrossRef - Cancer incidence and mortality and risk factors in member countries of the " Belt and Road " initiative
Baohua Wang, Fengdie He, Yanan Hu, Qiutong Wang, Dan Wang, Yuting Sha, Jing Wu
BMC Cancer.2022;[Epub] CrossRef - Geographical Differences in Kidney Cancer Outcomes of Patients Treated with Immunotherapy: A Systematic Review
Vinicius Knackfuss Gonçalves, Fernando Sabino Marques Monteiro, Antonia Angeli Gazola, Felipe Pizzolo, Júlia Elisa Hübner, Rodrigo Pellegrini, Alessandra Borba, André P. Fay
Kidney Cancer.2022; 6(2): 129. CrossRef - Global burden of disease study at the world health organization:
research methods for the most comprehensive global study of disease and
underlying health policies
Lee Smith, Jae Il Shin, Soo Young Hwang, Kalthoum Tizaoui, Elena Dragioti, Louis Jacob, Karel Kostev, Seung Won Lee, Ai Koyanagi
Life Cycle.2022;[Epub] CrossRef - Quality of care of peptic ulcer disease worldwide: A systematic analysis for the global burden of disease study 1990–2019
Mohsen Abbasi-Kangevari, Naser Ahmadi, Nima Fattahi, Negar Rezaei, Mohammad-Reza Malekpour, Seyyed-Hadi Ghamari, Sahar Saeedi Moghaddam, Sina Azadnajafabad, Zahra Esfahani, Ali-Asghar Kolahi, Shahin Roshani, Sahba Rezazadeh-Khadem, Fateme Gorgani, Seyyed
PLOS ONE.2022; 17(8): e0271284. CrossRef - A scoping review on population-centered indicators for cancer care continuum
Vasuki Rajaguru, Jieun Jang, Jeoung A. Kwon, Jae Hyun Kim, Jaeyong Shin, Mison Chun
Frontiers in Public Health.2022;[Epub] CrossRef - COMPARISON OF TEMPORAL TRENDS IN THE GLOBAL BURDEN OF TRACHEA, BRONCHIAL, AND LUNG CANCER
İskender ÇETİNTÜRK, Vahit YİĞİT
Giresun Üniversitesi İktisadi ve İdari Bilimler Dergisi.2022; 8(2): 254. CrossRef - ANALYSIS OF INCIDENCE AND MORTALITY OF GASTROINTESTINAL MALIGNANT TUMORS IN UKRAINE AND WORLDWIDE
Valeriy Zub
Acta Medica Leopoliensia.2022; 28(3-4): 22. CrossRef - Global, regional, national burden of breast cancer in 185 countries: evidence from GLOBOCAN 2018
Rajesh Sharma
Breast Cancer Research and Treatment.2021; 187(2): 557. CrossRef - A global, regional, and national survey on burden and Quality of Care Index (QCI) of hematologic malignancies; global burden of disease systematic analysis 1990–2017
Mohammad Keykhaei, Masood Masinaei, Esmaeil Mohammadi, Sina Azadnajafabad, Negar Rezaei, Sahar Saeedi Moghaddam, Nazila Rezaei, Maryam Nasserinejad, Mohsen Abbasi-Kangevari, Mohammad-Reza Malekpour, Seyyed-Hadi Ghamari, Rosa Haghshenas, Kamyar Koliji, Far
Experimental Hematology & Oncology.2021;[Epub] CrossRef - The global landscape of drug development of trastuzumab biosimilars
Dario Trapani, Giuseppe Curigliano
Journal of Cancer Policy.2021; 28: 100273. CrossRef - Trend analysis of cardiovascular disease mortality, incidence, and mortality-to-incidence ratio: results from global burden of disease study 2017
Maedeh Amini, Farid Zayeri, Masoud Salehi
BMC Public Health.2021;[Epub] CrossRef - Global, regional, and national burden and quality of care index (QCI) of thyroid cancer: A systematic analysis of the Global Burden of Disease Study 1990–2017
Sina Azadnajafabad, Sahar Saeedi Moghaddam, Esmaeil Mohammadi, Negar Rezaei, Erfan Ghasemi, Nima Fattahi, Arya Aminorroaya, Reza Azadnajafabad, Armin Aryannejad, Nazila Rezaei, Shohreh Naderimagham, Vahid Haghpanah, Ali H. Mokdad, Hossein Gharib, Farshad
Cancer Medicine.2021; 10(7): 2496. CrossRef - National and Subnational Incidence, Mortality, and Years of Life Lost Due to Breast Cancer in Iran: Trends and Age-Period-Cohort Analysis Since 1990
Bahar Ataeinia, Sahar Saeedi Moghaddam, Mahsima Shabani, Kimiya Gohari, Ali Sheidaei, Nazila Rezaei, Shohreh Naderimagham, Erfan Ghasemi, Mahtab Rouhifard Khalilabad, Shahin Roshani, Yosef Farzi, Farshad Farzadfar
Frontiers in Oncology.2021;[Epub] CrossRef - Honey and its protein components: Effects in the cancer immunology
Ivan Chan‐Zapata, Maira Rubí Segura‐Campos
Journal of Food Biochemistry.2021;[Epub] CrossRef - A Comparison of the United States and Austrian Healthcare Needs and Systems
Margaret McAlister, Joey D. Helton
INQUIRY: The Journal of Health Care Organization, Provision, and Financing.2021; 58: 004695802110001. CrossRef - Recent trend of androgen deprivation therapy in newly diagnosed prostate cancer patients: Comparing between high‐ and middle‐income Asian countries
Jasmine Lim, Mizuki Onozawa, Marniza Saad, Teng Aik Ong, Rohan Malek, Hideyuki Akaza
Cancer Science.2021; 112(6): 2071. CrossRef - A spatial assessment of prostate cancer mortality-to-incidence ratios among South Carolina veterans: 1999–2015
Peter Georgantopoulos, Jan M. Eberth, Bo Cai, Gowtham Rao, Charles L. Bennett, Christopher T. Emrich, Kathlyn S. Haddock, James R. Hébert
Annals of Epidemiology.2021; 59: 24. CrossRef - Assessment of quality benchmarks in adenoma detection in Mexico
Nancy E. Aguilar-Olivos, Ricardo Balanzá, Fernando Rojas-Mendoza, Rodrigo Soto-Solis, Mario A. Ballesteros-Amozurrutia, Norma González-Uribe, Justo A. Fernández-Rivero
Endoscopy International Open.2021; 09(06): E796. CrossRef - Favorable Lip and Oral Cancer Mortality-to-Incidence Ratios in Countries with High Human Development Index and Expenditures on Health
Wen-Wei Sung, Yong-Chen Hsu, Chen Dong, Ying-Ching Chen, Yu-Chi Chao, Chih-Jung Chen
International Journal of Environmental Research and Public Health.2021; 18(11): 6012. CrossRef - In prostatic transition zone lesions (PI-RADS v2.1): which subgroup should be biopsied?
Jihae An, Young Joong Kim, Jae Young Seo, Cheol Mog Hwang, Dong Hyun Oh, Mu-Sik Lee, Keum Won Kim
Egyptian Journal of Radiology and Nuclear Medicine.2021;[Epub] CrossRef - Improvement in the Mortality-to-Incidence Ratios for Gastric Cancer in Developed Countries With High Health Expenditures
Tzu-Wei Yang, Chi-Chih Wang, Wei-Cheng Hung, Yu-Hsiang Liu, Wen-Wei Sung, Ming-Chang Tsai
Frontiers in Public Health.2021;[Epub] CrossRef - Provider Barriers and Facilitators of Breast Cancer Guideline-Concordant Therapy Delivery in Botswana: A Consolidated Framework for Implementation Research Analysis
Tlotlo Ralefala, Lebogang Mokokwe, Swetha Jammalamadugu, Dumelang Legobere, Warona S. Motlhwa, Anthony A. Oyekunle, Surbhi Grover, Frances K. Barg, Lawrence N. Shulman, Yehoda M. Martei
The Oncologist.2021; 26(12): e2200. CrossRef - Improving Quality Indicator of Melanoma Management – Change of Melanoma Mortality-to-Incidence Rate Ratio Based on a Hungarian Nationwide Retrospective Study
Máté Várnai, Zoltán Kiss, Rolland Gyulai, Judit Oláh, Péter Holló, Gabriella Emri, András Csejtei, István Kenessey, Angéla Benedek, Zoltán Polányi, Zsófia Nagy-Erdei, Andrea Dániel, Kata Knollmajer, György Rokszin, Ibolya Fábián, Zsófia Barcza, Csaba Polg
Frontiers in Oncology.2021;[Epub] CrossRef - Cancer survival status among male population of Northeast India: A hospital based study
Manoj Kalita, JagannathDev Sharma, Debanjana Barman, Nizara Baishya, ChandiRam Kalita
Annals of Oncology Research and Therapy.2021; 1(1): 28. CrossRef - Global Burden of Head and Neck Cancer: Economic Consequences, Health, and the Role of Surgery
Rolvix H. Patterson, Victoria G. Fischman, Isaac Wasserman, Jennifer Siu, Mark G. Shrime, Johannes J. Fagan, Wayne Koch, Blake C. Alkire
Otolaryngology–Head and Neck Surgery.2020; 162(3): 296. CrossRef - Trends of National and Subnational Incidence of Childhood Cancer Groups in Iran: 1990–2016
Mahsima Shabani, Sahar Saeedi Moghaddam, Bahar Ataeinia, Nazila Rezaei, Farnam Mohebi, Bahram Mohajer, Kimiya Gohari, Ali Sheidaei, Farhad Pishgar, Moein Yoosefi, Farzad Kompani, Farshad Farzadfar
Frontiers in Oncology.2020;[Epub] CrossRef - Evaluation of the Cardiac Protection Conferred by Proanthocyanidins in Grape Seeds against Development of Ehrlich Solid Tumors in Mice
Maha Abdulrahman Aldubayan
BioMed Research International.2020; 2020: 1. CrossRef - The economic burden of cancer care for Syrian refugees: a population-based modelling study
Rima A Abdul-Khalek, Ping Guo, Forbes Sharp, Adrian Gheorghe, Omar Shamieh, Tezer Kutluk, Fouad Fouad, Adam Coutts, Ajay Aggarwal, Deborah Mukherji, Ghassan Abu-Sittah, Kalipso Chalkidou, Richard Sullivan
The Lancet Oncology.2020; 21(5): 637. CrossRef - Multiple Myeloma Incidence and Mortality Around the Globe; Interrelations Between Health Access and Quality, Economic Resources, and Patient Empowerment
Heinz Ludwig, Susie Novis Durie, Angela Meckl, Axel Hinke, Brian Durie
The Oncologist.2020; 25(9): e1406. CrossRef - Descriptive epidemiology of incidence and mortality of primary liver cancer in 185 countries: evidence from GLOBOCAN 2018
Rajesh Sharma
Japanese Journal of Clinical Oncology.2020; 50(12): 1370. CrossRef - Evaluating the Effect of Medicaid Expansion on Black/White Breast Cancer Mortality Disparities: A Difference-in-Difference Analysis
Jason Semprini, Olufunmilayo Olopade
JCO Global Oncology.2020; (6): 1178. CrossRef - State-Level Disparity in Lung Cancer Survival in the United States
Yu-Che Lee, Rafael A. Calderon-Candelario, Gregory E. Holt, Michael A. Campos, Mehdi Mirsaeidi
Frontiers in Oncology.2020;[Epub] CrossRef - Inequalities in the burden of female breast cancer in Brazil, 1990–2017
Maximiliano Ribeiro Guerra, Mário Círio Nogueira, Deborah Carvalho Malta, Camila Soares Lima Côrrea, Maria de Fatima Marinho de Souza, Maria Paula Curado, Mariana Santos Felisbino-Mendes, Meghan Mooney, Mohsen Naghavi, Maria Teresa Bustamante-Teixeira
Population Health Metrics.2020;[Epub] CrossRef - Disparities in mortality‐to‐incidence ratios by race/ethnicity for female breast cancer in New York City, 2002‐2016
Tamar B. Nobel, Charles K. Asumeng, John Jasek, Kellie C. Van Beck, Ruchi Mathur, Baozhen Qiao, Jennifer J. Brown
Cancer Medicine.2020; 9(21): 8226. CrossRef - Enhanced Cancer-targeted Drug Delivery Using Precoated Nanoparticles
Lu Yu, Mingyu Xu, Wenwen Xu, Wei Xiao, Xue-hua Jiang, Ling Wang, Huile Gao
Nano Letters.2020; 20(12): 8903. CrossRef - Oxaliplatin-Based Intra-arterial Chemotherapy in Colo-Rectal Cancer Liver Metastases: A Review from Pharmacology to Clinical Application
Girolamo Ranieri, Mariarita Laforgia, Patrizia Nardulli, Simona Ferraiuolo, Pasquale Molinari, Ilaria Marech, Cosmo Damiano Gadaleta
Cancers.2019; 11(2): 141. CrossRef - Melanoma prognosis in the United States: Identifying barriers for improved care
Zachary H. Hopkins, Christopher Moreno, Ryan Carlisle, Aaron M. Secrest
Journal of the American Academy of Dermatology.2019; 80(5): 1256. CrossRef - The Coincidence Between Increasing Age, Immunosuppression, and the Incidence of Patients With Glioblastoma
Erik Ladomersky, Denise M. Scholtens, Masha Kocherginsky, Elizabeth A. Hibler, Elizabeth T. Bartom, Sebastian Otto-Meyer, Lijie Zhai, Kristen L. Lauing, Jaehyuk Choi, Jeffrey A. Sosman, Jennifer D. Wu, Bin Zhang, Rimas V. Lukas, Derek A. Wainwright
Frontiers in Pharmacology.2019;[Epub] CrossRef - The Mortality-to-Incidence Ratio Is Not a Valid Proxy for Cancer Survival
Libby Ellis, Aurélien Belot, Bernard Rachet, Michel P. Coleman
Journal of Global Oncology.2019; (5): 1. CrossRef - National Cancer Mortality-to-Incidence Ratio (MIR) in Iran (2005 - 2014)
Atieh Akbari, Maryam Khayamzadeh, Reza Salmanian, Ali Ghanbari Motlagh, Gholamreza Roshandel, Maryam Nouri, Mohammad Esmaeil Akbari
International Journal of Cancer Management.2019;[Epub] CrossRef - The Association between Mortality-to-Incidence Ratios and Health Expenditures in Brain and Nervous System Cancers
Tsung-Han Lee, Wen-Wei Sung, Lung Chan, Hsiang-Lin Lee, Sung-Lang Chen, Yu-Hui Huang, Aij-Lie Kwan
International Journal of Environmental Research and Public Health.2019; 16(15): 2739. CrossRef - miR‐181a/b therapy in lung cancer: reality or myth?
Cornelia Braicu, Diana Gulei, Roxana Cojocneanu, Lajos Raduly, Ancuta Jurj, Erik Knutsen, George Adrian Calin, Ioana Berindan‐Neagoe
Molecular Oncology.2019; 13(1): 9. CrossRef - Breast Cancer: Reimbursement Policies and Adoption of New Therapeutic Agents by National Health Systems
Dario Trapani, Giuseppe Curigliano, Alexandru Eniu
Breast Care.2019; 14(6): 373. CrossRef - Epidemiology of prostate cancer in Asian countries
Takahiro Kimura, Shin Egawa
International Journal of Urology.2018; 25(6): 524. CrossRef - Cause-of-death statistics in 2016 in the Republic of Korea
Hyun-Young Shin, Ji-Youn Lee, Jee-Eun Kim, Seokmin Lee, Heejo Youn, Heyran Kim, Jeonghun Lee, Min Sim Park, Sun Huh
Journal of the Korean Medical Association.2018; 61(9): 573. CrossRef - Is mortality-to-incidence ratio associated with health disparity in pancreatic cancer? A cross-sectional database analysis of 57 countries
Hsiang-Lin Lee, Cheng-Ming Peng, Cheng-Yu Huang, Shin-Yi Wu, Ming-Chang Tsai, Chi-Chih Wang, Sung-Lang Chen, Chun-Che Lin, Chien-Ning Huang, Wen-Wei Sung
BMJ Open.2018; 8(7): e020618. CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite


