Articles
- Page Path
- HOME > Epidemiol Health > Volume 38; 2016 > Article
-
Original Article
Adjusting for reverse causation to estimate the effect of obesity on mortality after incident heart failure in the Atherosclerosis Risk in Communities (ARIC) study -
Maryam Shakiba1,2
 , Hamid Soori3
, Hamid Soori3 , Mohammad Ali Mansournia4
, Mohammad Ali Mansournia4 , Seyed Saeed Hashemi Nazari1
, Seyed Saeed Hashemi Nazari1 , Yahya Salimi5
, Yahya Salimi5
-
Epidemiol Health 2016;38:e2016025.
DOI: https://doi.org/10.4178/epih.e2016025
Published online: June 4, 2016
1Department of Epidemiology, School of Public Health, Shahid Beheshti University of Medical Sciences, Tehran, Iran
2Interventional Cardiovascular Research Center, Guilan University of Medical Sciences, Rasht, Iran
3Safety Promotion and Injury Prevention Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran
4Department of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
5Department of Epidemiology and Biostatistics, School of Public Health, Kermanshah University of Medical Sciences, Kermanshah, Iran
- Correspondence: Hamid Soori Safety Promotion and Injury Prevention Research Center, Shahid Beheshti University of Medical Sciences, Parvaneh St., Velenjak Area, Chamran Highway, Tehran, Iran Tel/Fax: +98-212243998, E-mail: hsoori@yahoo.com
©2016, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES:
- The lower mortality rate of obese patients with heart failure (HF) has been partly attributed to reverse causation bias due to weight loss caused by disease. Using data about weight both before and after HF, this study aimed to adjust for reverse causation and examine the association of obesity both before and after HF with mortality.
-
METHODS:
- Using the Atherosclerosis Risk in Communities (ARIC) study, 308 patients with data available from before and after the incidence of HF were included. Pre-morbid and post-morbid obesity were defined based on body mass index measurements at least three months before and after incident HF. The associations of pre-morbid and post-morbid obesity and weight change with survival after HF were evaluated using a Cox proportional hazard model.
-
RESULTS:
- Pre-morbid obesity was associated with higher mortality (hazard ratio [HR], 1.61; 95% confidence interval [CI], 1.04 to 2.49) but post-morbid obesity was associated with increased survival (HR, 0.57; 95% CI, 0.37 to 0.88). Adjusting for weight change due to disease as a confounder of the obesity-mortality relationship resulted in the absence of any significant associations between post-morbid obesity and mortality.
-
CONCLUSIONS:
- This study demonstrated that controlling for reverse causality by adjusting for the confounder of weight change may remove or reverse the protective effect of obesity on mortality among patients with incident HF.
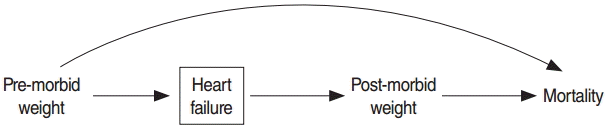
- Despite the fact that obesity is positively related to all-cause mortality in the general population [1,2], the “obesity paradox,” defined as a lower mortality rate of obese and overweight individuals compared to normal-weight individuals, has been recognized for various chronic diseases [3-5], including chronic heart failure (HF) [6,7]. Among some plausible explanations that have been presented, some authors have attributed this paradox to a higher metabolic reserve putatively present among obese patients [8,9]. In contrast, other authors have explained the obesity paradox as a result of bias due to reverse causation [10-12]. Briefly, this explanation proposes that low weight is a consequence of pre-existing illness resulting in weight loss, which can distort the true relationship between body weight and the risk of death [13-16]. Some strategies for avoiding this form of bias have been implemented in previous studies, such as performing research in occupational cohorts, restricting samples to healthy individuals who are less likely to have chronic diseases, following up for a long period of time, excluding early deaths that may have occurred due to pre-existing illness, and/or using maximum lifetime body mass index (BMI) [1,13,17,18]. All of these approaches have been implemented in the general population, but due to the limited size of available patients, these methods may have limited applicability for evaluating the obesity-mortality relationship in patients with chronic diseases. In this regard, pre-existing illness is also a confounding variable since it affects both body weight (through weight loss) and future mortality [15,19,20]. This study proposed that weight change due to HF was a variable that could serve as a confounder for the obesity-mortality relationship and that adjusting for it may eliminate the reverse causation bias due to pre-existing disease.
- In light of the discordant opinions regarding the obesity-mortality relationship, we conducted an analysis using the Atherosclerosis Risk in Communities (ARIC) study to investigate the association between obesity and future mortality in patients with incident HF. One aim of this study was to examine the effect of reverse causation by controlling for weight change as confounder. The hypothesis was that if weight loss distorts the true association between body weight and mortality, the obesity-mortality relationship will disappear as a result of adjusting for weight change. The other aim of this study was to estimate the association of obesity both before and after the development of HF with mortality. If greater metabolic reserve contributes to lower mortality, then obesity before the development of HF would be associated with better survival.
INTRODUCTION
- Study population
- The ARIC study was a community-based prospective cohort study designed to investigate the causes of atherosclerosis. The study recruited 15,792 adults aged 45 years to 64 years at baseline who resided in four US communities. The first examinations of participants (visit 1) took place during 1987 to 1989, followed by three visits at 3-year intervals. The follow-up rates for the first, second, and third intervals were 91%, 82%, and 74%, respectively. At each visit, participants responded to an interviewer-administered questionnaire and underwent an extensive physical examination [21]. The current study was restricted to patients with incident HF through December 31, 2009. Among 2,169 patients with incident HF, 1,861 cases were excluded because HF developed after the fourth examination, meaning that no data were available after the development of disease. Therefore, the present study included 308 patients with data available both before and after the incidence of HF. The study sample had a 0% prevalence of HF at baseline. This study was approved by the institutional review board of the Shahid Beheshti University of Medical Sciences.
- Determination of heart failure and mortality outcomes
- Incident HF was defined as death or hospitalization with an International Classification of Diseases, 9th revision (ICD-9) code for HF (i.e., any variation of 410) given the absence of prevalent HF at visit 1. HF cases were determined through annual telephone interviews regarding their experiences of hospitalization, reviewing hospital discharge lists, and a survey of the national death index and death certificate files. The latter two methods were performed through community-wide surveillance of the communities involved in the ARIC study. The response rate for telephone interviews was 93% to 96%. Mortality outcomes were evaluated in the same way.
- Covariates
- For this study, demographic variables including sex, race (black or white), and education level (basic or no formal education, intermediate, and advanced education) were determined at visit 1. The data before and after the development of HF were considered to define pre-morbid and post-morbid variables. Premorbid variables were based the measurement of covariates ≥3 months before incident HF and post-morbid variables were defined as measurements of covariates ≥3 months after incident HF. On this basis, pre-morbid and post-morbid values for comorbidities including a history of cancer, coronary heart disease, stroke and chronic lung disease, as well as covariates including smoking and drinking status, diabetes mellitus, hypertension, and total serum cholesterol were obtained. Similarly, pre-morbid and post-morbid obesity were defined using BMI measurements from study visits occurring ≥3 months prior and ≥3 months after the date of incident HF, respectively. Due to the small sample of available patients, we categorized BMI into non-obese (BMI <30 kg/m2) and obese (BMI ≥30 kg/m2). Weight change was defined as percentage change in BMI and calculated as the change in BMI value divided by the original (pre-HF) values of BMI multiplied by 100. Weight change was then categorized based on a priori cutpoint [10]. A weight change of ≤-6% was classified as weight loss, a change of -6% to 6% was classified as stable weight, and a change of ≥6% was classified as weight gain. Smoking status was defined as positive if the subjects ever smoked or smoked cigarettes at the time of the study. Drinking status was considered as positive if subjects regularly drank at the time of the study or had ever consumed alcoholic beverages. Hypertension was defined as a systolic blood pressure ≥90 mmHg, a diastolic blood pressure ≥140 mmHg, or the use of medication for high blood pressure in the previous two weeks. Diabetes mellitus was defined as a non-fasting blood glucose ≥200 mg/dL, a fasting blood glucose level ≥126 mg/dL, or the use of diabetes medication in the previous two weeks. Fasting blood glucose and total serum cholesterol were measured after overnight fasting. Cholesterol level was categorized using the common cutpoint of ≥240 mg/dL.
- Statistical analysis
- Baseline data were described as mean (standard deviation, SD) or frequency (percentage) and compared using the t-test or the chi-square test. The pre-morbid and post-morbid measurements of risk factors were compared using the paired t-test or the McNemar test. Correlations between pre-morbid and post-morbid covariates were evaluated using the Spearman test. In the multivariate adjusted model, pre-morbid covariates were dropped if they were highly correlated with post-morbid covariate values (r≥0.80). The endpoint of interest was time to all-cause mortality after incident HF. A Cox proportional hazard model with age as the underlying time scale was used to examine the effect of pre-morbid and post-morbid obesity and other covariates on survival. The proportionality assumption was assessed using a graphical approach and testing the slope of Schoenfeld residuals. The Cox model used the date of birth as the origin of the time scale. In order to calculate weight change as a confounder, we used each participant’s weight before the potential onset of disease as their original weight. Weight change naturally occurs before post-morbid obesity, so it was not an intermediate variable between the exposure of interest and outcome. All statistical analyses were performed using Stata version 13.0 (StataCorp, College Station, TX, USA).
MATERIALS AND METHODS
- The median and interquartile range of the duration from the previous visit to incident HF was 24.5 (14 to 31) months and from incident HF to the post-visit follow-up was 17 (9 to 27) months. Among 308 patients with incident HF and data available for the pre-morbid and post-morbid covariates, 47% were obese pre-morbidity, 61% were male, 33% were black, and 44% had a basic education. The mean age at the incidence of HF was 61 years old (SD, 5.9 years). A total of 3,027 person-years were incorporated into this study, during which 231 deaths occurred. The median follow-up was 9.8 years and the death rate was 76 per 1,000 person-years. Table 1 presents the pre-morbid and post-morbid characteristics of patients with incident HF according to pre-morbid and post-morbid obesity. Pre-morbid obese patients were younger, more likely to be black, and had a higher percentage of diabetes and hypertension. In contrast, the percentage of smoking status was lower in obese patients than in non-obese patients. No significant differences were found in education levels, drinking status, high cholesterol value, history of coronary heart disease, cancer, stroke, chronic lung disease, and weight change categories. Post-morbidity, the overall prevalence of smoking and drinking decreased. Post-morbid obese patients also had a lower prevalence of drinking and smoking than non-obese patients. However, the prevalence of diabetes and hypertension increased, and these conditions were more common in post-morbid obese patients than in non-obese patients. The mean percentage of weight change was -2.71% (minimum, -34%; maximum, 90%). The percentages of patients who experienced weight loss and weight gain were 30% and 20%, respectively. A significant difference was found in the death rate among the three weight change categories (p=0.02), with higher crude death rates in patients who experienced weight loss (95 per 1,000 person-years), compared to those with a stable weight and those who underwent weight gain (70 and 67 per 1,000 person-years, respectively).
- A multivariate adjusted model was used to determine the associations of pre-morbid and post-morbid obesity with mortality. Based on subject matter knowledge and the graphical representation of variables as a directed acyclic graph in Figure 1, weight before the development of HF may have confounded the relation between post-morbid weight and mortality. Weight after the development of HF was also a strong mediator for the effect of pre-morbid obesity on mortality. Therefore, in order to estimate the direct effect of pre-morbid obesity, we adjusted for post-morbid obesity and other covariates. The results of the multivariate adjusted model are presented in Table 2. It was found that the hazard ratio (HR) of mortality in pre-morbid obese patients was significantly higher than in non-obese patients (HR, 1.61; 95% confidence interval [CI], 1.04 to 2.49), while post-morbid obesity was associated with a reduced risk of mortality (HR, 0.57; 95%CI, 0.37 to 0.88). In this model, post-morbid diabetes also significantly increased the risk of mortality. Pre-morbid diabetes was dropped from the model due to its high correlation (r=0.85) with post-morbid diabetes.
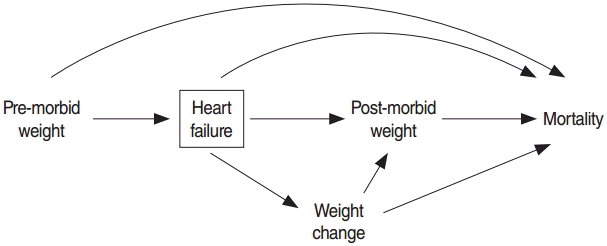
- According to the reverse causation hypothesis, as presented in Figure 2, weight change due to disease may confound the association of post-morbid obesity and mortality. Table 3 shows the distribution of weight change by post-morbid BMI category. A strong association was found between weight change and post-morbid obesity status, such that 40% of non-obese patients experienced weight loss, in contrast to only 18% of obese patients. The results of the univariate Cox model showed that a strong inverse association was present between weight change and mortality. The risk of death decreased by 3% with each 1-unit increment of the percentage change in BMI (HR, 0.97; 95% CI, 0.96 to 0.98). Table 4 shows the effect of post-morbid obesity on all-cause mortality adjusting for weight change and other pre-morbid and post-morbid covariates. In this model, post-morbid obesity was no longer a significant protective factor. The adjusted HR for weight change showed that patients who experienced weight loss had a 42% higher all-cause mortality (95% CI, 1.02 to 1.97), whereas patients who experienced weight gain had a 33% reduction in all-cause mortality compared to stable-weight patients (Table 4).
RESULTS
- In this study, reverse causation was revisited and evaluated using a new approach that treated weight change due to disease as a confounder. Our results demonstrate that reverse causation may result in the protective effect of post-morbid obesity on all-cause mortality in patients with incident HF. This conclusion is supported by the strong association we found between weight change due to disease and post-morbid obesity and the association of weight loss with all-cause mortality. Therefore, mortality in obese patients may be reduced due to their lower likelihood of having experienced weight loss. We used a relative measure for weight change in which all patients who lost more than 6% of their original BMI were categorized as experiencing weight loss, irrespective of their original weight.
- The issue of reverse causation as a form of bias occurring in studies of obesity and mortality has generally been accounted for in the general population [1,13,17,18]. To our knowledge, though, this is the first study to adjust for reverse causation in patients with HF. In this study, we hypothesized that weight change due to HF could serve as a confounder for the obesity-mortality relationship. The main strength of this study was that weight change was calculated from the weight before any potential onset of disease. Hence, any changes in weight could attributed to the disease condition. Furthermore, since some cases of weight loss may have been caused by a reduction in edema that could have been present in the early stages of HF, calculating weight changes based on the weight before the development of disease would better reflect weight changes as a result of disease. The results of the current study showed an increased risk of all-cause mortality for patients who experienced weight loss compared to stable-weight patients, independently of post-morbid obesity and other covariates. This finding is consistent with those of previous studies that have found strong independent effects of weight loss during the course of disease on mortality [8,10-12,22-25]. Our results support the importance of reverse causation as a plausible explanation for the lower mortality rates of obese patients. After adjustment for weight changes due to disease, the significant protective association of obesity on mortality disappeared.
- In this study, pre-morbid obese patients who eventually developed HF had a higher risk of mortality than pre-morbid non-obese patients. This finding is in contrast with a previous report that found better survival rates among pre-morbid obese patients [26]. The main shortcoming of the previous study is that it did not consider the effect of the post-morbid weight as an indirect effect of pre-morbid obesity on mortality. The development of edema as a result of salt and water retention in the early stages of HF and wasting due to cardiac cachexia in the later stage influence weight after the incidence of HF. Therefore, in order to estimate the direct effect of pre-morbid weight, weight after disease should be adjusted for. Our finding regarding the higher mortality rate of pre-morbid obese patients does not support the putative protective effect of higher metabolic reserve in obese patients. However, our result demonstrated support for the obesity paradox by documenting better survival associated with post-morbid obesity, disregarding the effect of weight change as a confounder.
- Although the term “obesity paradox” is accepted by many scientific communities and listed in some guidelines for cardiovascular disease, it seems that some methodological flaws still persist in estimating the effect of obesity among patients suffering from chronic diseases [27]. This study highlighted the effect of weight change as an important confounder for the obesity-mortality relationship. In fact, the category of non-obese patients after disease development may include many patients who have experienced weight loss and have a higher risk of mortality, which erroneously makes obesity appear to be a protective factor [9,15].
- This study suffers from some limitations. The identification of incident HF cases was only based on ICD-9 codes for hospitalizations and deaths due to HF, which were not validated, while outpatients with HF were included through responses from physicians using a form. However, the validation of ICD-9 codes by a subsequent medical record review following the ARIC criteria in the ARIC community surveillance study showed a sensitivity of 95% and a positive predictive value of 85%, which are both higher than the Framingham criteria [28]. Additionally, due to the small number of participants with data available before and after incident HF, pre-morbid and post-morbid variables were defined as events occurring at an interval of at least three months. Furthermore, BMI was categorized into two categories, comparing obese and non-obese patients. Therefore, we could not assess differences among all BMI categories, including overweight and underweight. Finally, this study has the inherent limitation of residual confounding due to unmeasured covariates, such as nutritional status and physical fitness, which could make associations a poor estimate of causal effects.
- In this study, obese patients who developed HF had a higher risk of mortality than non-obese patients. Thus, greater metabolic reserve did not serve as a protective factor against mortality among obese patients. Post-morbid obesity was not a significant prognostic factor for mortality when the relationship was adjusted for weight change due to HF. Reverse causation may be a plausible explanation for the lower mortality risk of post-morbid obese patients.
DISCUSSION
ACKNOWLEDGEMENTS

|
Pre-morbid obesity |
p-value1 |
Post-morbid obesity |
p-value | p-value2 (within group) | |||
|---|---|---|---|---|---|---|---|
| No (n = 164) | Yes (n = 144) | No (n = 178) | Yes (n = 130) | ||||
| Age at baseline (yr) | 62 (0.4) | 60 (0.5) | 0.001 | ||||
| Sex (male) | 110 (67) | 77 (53) | 0.01 | 111 (62) | 76 (58) | 0.45 | |
| Race (black ) | 44 (26) | 59 (41) | 0.005 | 58 (32) | 45 (34) | 0.71 | |
| Education level | 0.14 | 0.83 | |||||
| Basic | 58 (35) | 66 (46) | 70 (39) | 54 (41) | |||
| Intermediate | 64 (39) | 51 (35) | 66 (37) | 49 (38) | |||
| Advanced | 42 (26) | 27 (19) | 42 (24) | 27 (21) | |||
| Smoking status | 64 (39) | 35 (25) | 0.007 | 42 (24) | 13 (11) | 0.008 | 0.001 |
| Drinking status | 78 (48) | 54 (38) | 0.09 | 66 (38) | 26 (20) | 0.009 | 0.001 |
| Diabetes mellitus | 45 (27) | 64 (46) | 0.001 | 55 (32) | 66 (58) | 0.001 | 0.001 |
| Hypertension | 88 (54) | 98 (71) | 0.003 | 110 (64) | 87 (76) | 0.03 | 0.003 |
| Total cholesterol (≥240 mg/dL) | 41 (25) | 32 (23) | 0.74 | 28 (16) | 19 (17) | 0.83 | 0.002 |
| History of coronary heart disease | 49 (31) | 39 (28) | 0.58 | 106 (61) | 57 (51) | 0.11 | |
| History of cancer | 9 (5) | 8 (6) | 0.94 | 6 (3) | 2 (2) | 0.40 | |
| History of stroke | 10 (6) | 8 (6) | 0.85 | 18 (10) | 13 (11) | 0.73 | |
| History of chronic lung disease | 11 (7) | 7 (5) | 0.50 | 15 (9) | 15 (13) | 0.20 | |
| Weight change | 0.67 | ||||||
| Weight loss | 47 (29) | 47 (32) | |||||
| Stable weight | 81 (49) | 70 (49) | |||||
| Weight gain | 36 (22) | 27 (19) | |||||
| Covariates | No. of events (person-years) | Crude | p-value | Adjusted1 | p-value |
|---|---|---|---|---|---|
| Sex | 0.85 | 0.15 | |||
| Female | 88 (1,193) | 1.00 (reference) | 1.00 (reference) | ||
| Male | 143 (1,830) | 0.97 (0.74, 1.27) | 1.28 (0.91, 1.79) | ||
| Race | 0.001 | 0.16 | |||
| Black | 88 (872) | 1.00 (reference) | 1.00 (reference) | ||
| White | 143 (2,155) | 0.59 (0.45, 0.78) | 0.76 (0.52, 1.11) | ||
| Pre-morbid obesity | 107 (1,408) | 1.12 (0.86, 1.45) | 0.41 | 1.61 (1.04, 2.49) | 0.03 |
| Post-morbid obesity | 88 (1,269) | 0.89 (0.68, 1.16) | 0.39 | 0.57 (0.37, 0.88) | 0.01 |
| Pre-morbid smoking | 83 (966) | 1.36 (1.03, 1.79) | 0.03 | 1.13 (0.75, 1.70) | 0.56 |
| Post-morbid smoking | 50 (509) | 1.79 (1.29, 2.49) | 0.001 | 1.59 (0.99, 2.57) | 0.05 |
| Pre-morbid drinking | 92 (1,339) | 0.83 (0.64, 1.09) | 0.18 | 1.02 (0.70, 1.49) | 0.90 |
| Post-morbid drinking | 65 (987) | 0.81 (0.60, 1.09) | 0.17 | 0.89 (0.59, 1.35) | 0.60 |
| Post-morbid diabetes | 103 (1,064) | 1.80 (1.37, 2.37) | 0.001 | 1.78 (1.28, 2.47) | 0.001 |
| Pre-morbid hypertension | 146 (1,665) | 1.43 (1.09, 1.88) | 0.01 | 1.12 (0.78, 1.62) | 0.53 |
| Post-morbid hypertension | 152 (1,881) | 1.34 (0.99, 1.81) | 0.06 | 0.94 (0.62, 1.41) | 0.75 |
| Pre-morbid (cholesterol ≥240 mg/dL) | 59 (652) | 1.44 (1.06, 1.94) | 0.02 | 1.32 (0.91, 1.98) | 0.14 |
| Post-morbid (cholesterol ≥240 mg/dL) | 40 (439) | 1.31 (0.93, 1.85) | 0.12 | 0.85 (0.54, 1.34) | 0.49 |
| Non-obese (n = 178) | Obese (n = 130) | p-value | |
|---|---|---|---|
| Weight loss | 71 (40) | 23 (18) | 0.001 |
| Stable weight | 85 (48) | 66 (51) | |
| Weight gain | 22 (12) | 41(31) |
| Covariates | No. of events (person-years) | Crude | p-value | Adjusted1 | p-value |
|---|---|---|---|---|---|
| Weight change | |||||
| Stable weight | 108 (1,549) | 1.00 (reference) | 1.00 (reference) | ||
| Weight loss | 80 (833) | 1.43 (1.07, 1.91) | 0.01 | 1.42 (1.02, 1.97) | 0.03 |
| Weight gain | 43 (644) | 0.93 (0.65, 1.33) | 0.69 | 0.67 (0.41, 1.09) | 0.11 |
| Post-morbid obesity | 88 (1,269) | 0.89 (0.69, 1.17) | 0.69 | 0.80 (0.57, 1.12) | 0.19 |
- 1. Berrington de Gonzalez A, Hartge P, Cerhan JR, Flint AJ, Hannan L, MacInnis RJ, et al. Body-mass index and mortality among 1.46 million white adults. N Engl J Med 2010;363:2211-2219.ArticlePubMedPMC
- 2. Flegal KM, Kit BK, Orpana H, Graubard BI. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. JAMA 2013;309:71-82.ArticlePubMedPMC
- 3. Uretsky S, Messerli FH, Bangalore S, Champion A, Cooper-Dehoff RM, Zhou Q, et al. Obesity paradox in patients with hypertension and coronary artery disease. Am J Med 2007;120:863-870.ArticlePubMed
- 4. Sharma A, Lavie CJ, Borer JS, Vallakati A, Goel S, Lopez-Jimenez F, et al. Meta-analysis of the relation of body mass index to all-cause and cardiovascular mortality and hospitalization in patients with chronic heart failure. Am J Cardiol 2015;115:1428-1434.ArticlePubMed
- 5. Oreopoulos A, Padwal R, Kalantar-Zadeh K, Fonarow GC, Norris CM, McAlister FA. Body mass index and mortality in heart failure: a meta-analysis. Am Heart J 2008;156:13-22.ArticlePubMed
- 6. Shah R, Gayat E, Januzzi JL Jr, Sato N, Cohen-Solal A, diSomma S, et al. Body mass index and mortality in acutely decompensated heart failure across the world: a global obesity paradox. J Am Coll Cardiol 2014;63:778-785.ArticlePubMed
- 7. Kenchaiah S, Pocock SJ, Wang D, Finn PV, Zornoff LA, Skali H, et al. Body mass index and prognosis in patients with chronic heart failure: insights from the candesartan in heart failure: assessment of reduction in mortality and morbidity (CHARM) program. Circulation 2007;116:627-636.ArticlePubMed
- 8. Anker SD, Ponikowski P, Varney S, Chua TP, Clark AL, Webb-Peploe KM, et al. Wasting as independent risk factor for mortality in chronic heart failure. Lancet 1997;349:1050-1053.ArticlePubMed
- 9. Banack HR, Kaufman JS. The obesity paradox: understanding the effect of obesity on mortality among individuals with cardiovascular disease. Prev Med 2014;62:96-102.ArticlePubMed
- 10. Anker SD, Negassa A, Coats AJ, Afzal R, Poole-Wilson PA, Cohn JN, et al. Prognostic importance of weight loss in chronic heart failure and the effect of treatment with angiotensin-converting-enzyme inhibitors: an observational study. Lancet 2003;361:1077-1083.ArticlePubMed
- 11. Doehner W, Erdmann E, Cairns R, Clark AL, Dormandy JA, Ferrannini E, et al. Inverse relation of body weight and weight change with mortality and morbidity in patients with type 2 diabetes and cardiovascular co-morbidity: an analysis of the PROactive study population. Int J Cardiol 2012;162:20-26.ArticlePubMed
- 12. Pocock SJ, McMurray JJ, Dobson J, Yusuf S, Granger CB, Michelson EL, et al. Weight loss and mortality risk in patients with chronic heart failure in the candesartan in heart failure: assessment of reduction in mortality and morbidity (CHARM) programme. Eur Heart J 2008;29:2641-2650.ArticlePubMed
- 13. Adams KF, Schatzkin A, Harris TB, Kipnis V, Mouw T, Ballard-Barbash R, et al. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N Engl J Med 2006;355:763-778.Article
- 14. Flanders WD, Augestad LB. Adjusting for reverse causality in the relationship between obesity and mortality. Int J Obes (Lond) 2008;32 Suppl 3:S42-S46.Article
- 15. Flegal KM, Graubard BI, Williamson DF, Cooper RS. Reverse causation and illness-related weight loss in observational studies of body weight and mortality. Am J Epidemiol 2011;173:1-9.ArticlePubMed
- 16. Janssen I, Bacon E. Effect of current and midlife obesity status on mortality risk in the elderly. Obesity (Silver Spring) 2008;16:2504-2509.ArticlePubMed
- 17. Lawlor DA, Hart CL, Hole DJ, Davey Smith G. Reverse causality and confounding and the associations of overweight and obesity with mortality. Obesity (Silver Spring) 2006;14:2294-2304.ArticlePubMed
- 18. Stokes A. Using maximum weight to redefine body mass index categories in studies of the mortality risks of obesity. Popul Health Metr 2014;12:6.ArticlePubMedPMCPDF
- 19. Lewis CE, McTigue KM, Burke LE, Poirier P, Eckel RH, Howard BV, et al. Mortality, health outcomes, and body mass index in the overweight range: a science advisory from the American Heart Association. Circulation 2009;119:3263-3271.ArticlePubMed
- 20. Robins JM. Causal models for estimating the effects of weight gain on mortality. Int J Obes (Lond) 2008;32 Suppl 3:S15-S41.Article
- 21. ARIC Investigators. The Atherosclerosis Risk in Communities (ARIC) study: design and objectives. Am J Epidemiol 1989;129:687-702.PubMed
- 22. Lavie CJ, Milani RV, Artham SM, Patel DA, Ventura HO. The obesity paradox, weight loss, and coronary disease. Am J Med 2009;122:1106-1114.ArticlePubMed
- 23. Strandberg TE, Strandberg AY, Salomaa VV, Pitkälä KH, Tilvis RS, Sirola J, et al. Explaining the obesity paradox: cardiovascular risk, weight change, and mortality during long-term follow-up in men. Eur Heart J 2009;30:1720-1727.ArticlePubMed
- 24. Myers J, Lata K, Chowdhury S, McAuley P, Jain N, Froelicher V. The obesity paradox and weight loss. Am J Med 2011;124:924-930.ArticlePubMed
- 25. Kennedy LM, Dickstein K, Anker SD, James M, Cook TJ, Kristianson K, et al. Weight-change as a prognostic marker in 12 550 patients following acute myocardial infarction or with stable coronary artery disease. Eur Heart J 2006;27:2755-2762.ArticlePubMed
- 26. Khalid U, Ather S, Bavishi C, Chan W, Loehr LR, Wruck LM, et al. Pre-morbid body mass index and mortality after incident heart failure: the ARIC study. J Am Coll Cardiol 2014;64:2743-2749.ArticlePubMedPMC
- 27. Ferreira I, Stehouwer CD. Obesity paradox or inappropriate study designs? Time for life-course epidemiology. J Hypertens 2012;30:2271-2275.ArticlePubMed
- 28. Rosamond WD, Chang PP, Baggett C, Johnson A, Bertoni AG, Shahar E, et al. Classification of heart failure in the Atherosclerosis Risk in Communities (ARIC) study: a comparison of diagnostic criteria. Circ Heart Fail 2012;5:152-159.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Prevalence and clinical characteristics of diabetic cardiomyopathy in patients with acute heart failure
Kenichi Matsushita, Kazumasa Harada, Takashi Kohno, Hiroki Nakano, Daisuke Kitano, Junya Matsuda, Makoto Takei, Hideaki Yoshino, Takeshi Yamamoto, Ken Nagao, Morimasa Takayama
Nutrition, Metabolism and Cardiovascular Diseases.2024; 34(5): 1325. CrossRef - Impact of body mass index on cardiac adrenergic derangement in heart failure patients: a 123I-mIBG imaging study
Klara Komici, Leonardo Bencivenga, Stefania Paolillo, Paola Gargiulo, Roberto Formisano, Roberta Assante, Carmela Nappi, Fabio Marsico, Adriana D’Antonio, Giovanni De Simini, Antonio Cittadini, Dino Franco Vitale, Alberto Cuocolo, Pasquale Perrone Filardi
European Journal of Nuclear Medicine and Molecular Imaging.2020; 47(7): 1713. CrossRef - Impact of prior bariatric surgery on outcomes of hospitalized patients with heart failure: a population-based study
Hedong Han, Tiantian Zhu, Yibin Guo, Yiming Ruan, Eyal Herzog, Jia He
Surgery for Obesity and Related Diseases.2019; 15(3): 469. CrossRef - Body mass index and all-cause mortality in patients with atrial fibrillation: insights from the China atrial fibrillation registry study
Lu Wang, Xin Du, Jian-Zeng Dong, Wen-Na Liu, Ying-Chun Zhou, Song-Nan Li, Xue-Yuan Guo, Chen-Xi Jiang, Rong-Hui Yu, Cai-Hua Sang, Ri-Bo Tang, De-Yong Long, Nian Liu, Rong Bai, Laurent Macle, Chang-Sheng Ma
Clinical Research in Cardiology.2019; 108(12): 1371. CrossRef - The Effects of Reverse Causality and Selective Attrition on the Relationship Between Body Mass Index and Mortality in Postmenopausal Women
Hailey R Banack, Jennifer W Bea, Jay S Kaufman, Andrew Stokes, Candyce H Kroenke, Marcia L Stefanick, Shirley A Beresford, Chloe E Bird, Lorena Garcia, Robert Wallace, Robert A Wild, Bette Caan, Jean Wactawski-Wende
American Journal of Epidemiology.2019; 188(10): 1838. CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite