Articles
- Page Path
- HOME > Epidemiol Health > Volume 34; 2012 > Article
-
Original Article
Case Series ofMycobacterium Abscessus Infections Associated with a Trigger Point Injection and Epidural Block at a Rural Clinic - Jun Young Song1,2, Jung Bin Son1, Min Ki Lee2, Jin Gwack3, Kil Soo Lee4, Ji Young Park5
-
Epidemiol Health 2012;34:e2012001.
DOI: https://doi.org/10.4178/epih/e2012001
Published online: January 25, 2012
1Epidemic Intelligence Service, Division of Public Health Administration, Gyeongnam Provincial Office, Changwon, Korea.
2Department of Internal Medicine, Pusan National University School of Medicine and Medical Research Institute, Busan, Korea.
3Division of Epidemic Intelligence Service, Korea Centers for Disease Control and Prevention, Osong, Korea.
4Division of Bacterial Respiratory Infections, Center for Infectious Diseases, National Institute of Health, Korea Centers for Disease Control and Prevention, Seoul, Korea.
5Department of Internal Medicine, Kosin University College of Medicine, Busan, Korea.
- Correspondence: Ji Young Park, MD. Department of Internal Medicine, Kosin University College of Medicine, 34-1 Amnam-dong, Seo-gu, Busan 158-710, Korea. Tel: +82-51-990-6716, Fax: +82-51-990-3141, aiteite1@naver.com
© 2012, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 16,851 Views
- 109 Download
- 9 Crossref
Abstract
-
OBJECTIVES
- The aim of this report is to investigate Mycobacterium abscessus infections at a rural clinic and carry out a surveillance program to determine the extent and source of these infections.
-
METHODS
- The authors conducted an active surveillance investigation of 36 patients who had visited the clinic since 1 July 2008. Clinical specimens were collected from the patients and an envirnmental investigation. Pulsed-field gel elctrophoresis (PFGE) was performed for comparing with M. abscessus isolates from the patients.
-
RESULTS
- Six specimens were obtained from the 6 patients respectively and 22 environmental samples were obtained. M. abscessus was isolated from the wounds of two patients, and various nosocomial pathogens, but not M. abscessus, were isolated from the surrounding environment. Two strains of M. abscessus from patients were identical as a result of PFGE.
-
CONCLUSION
- Infection control education including proper hand hygiene should be emphasized for physicians performing invasive procedures. There also needs to be more attention for invasive procedures management, including trigger point injection and epidural block in rural clinics.
- Nontuberculous mycobacteria (NTM) are found in the natural environment throughout the world and include at least 65 different species [1]. NTM exist widely in soil and water and are known to colonize and cause disease in a broad range of animal species. There is sufficient evidence that nosocomial transmission of these organisms is increasing [2,3]. Mycobacterium abscessus is an acid-fast bacillus (AFB) classified as a pathogenic "rapid growing" strain of NTM [2,4]. M. abscessus was first reported by Moore and Frerichs in 1953 [5]. Although M. abscessus is an uncommon causative pathogen of human disease, it can cause skin and soft tissue infections (SSTI) on damaged skin lesions following inoculation, minor trauma or surgery [5,6].
- Trigger point injection (TPI) is widely used for treatment by some physicians to reduce myofascial pain and headache that is unresponsive to medical treatments in Korea [7,8]. Trigger points may irritate the nerves around them and cause referred pain, or pain that is felt in another part of the body. Lidocaine or steroid mixed normal saline injection with a 24 or 26 gauge needle to the trigger point helps to reduce pain. Many surveys regarding the safety of this procedure have been conducted but the effectiveness of TPI is controversial for a decade [9].
- There have been few reports of M. abscessus outbreak associated with TPI [10-12] and/or epidural block (EB) procedures over the past years. The aim of this study was to investigate M. abscessus infections at a rural clinic and carry out a surveillance program to determine the extent and source of these infections.
INTRODUCTION
- Outbreak recognition
- On 2 March 2009, a patient (index case) who was referred from a local clinic in Ha-dong-gun province, was diagnosed with an NTM infection in hospital A and was reported to the Korea Centers for Disease Control and Prevention (KCDC). He had multiple nodules and abscesses following EB procedure at a rural clinic in mid-October 2008. The lesions were initially treated with antibiotics, abscess incision and drainage, without improvement.
- On 9 March 2009, the team from KCDC, the public health center and the provincial government performed an epidemiological investigation at the clinic in Ha-dong-gun province and interviewed the physician who performed the TPI and/or EB procedures. In the field, our team was notified that similar patients had been treated in another hospital B. These events prompted the initiation of an epidemiological study to investigate the outbreak.
- From 1 July 2008 to 28 February 2009, 496 patients had visited the clinic in Ha-dong-gun province. We attempted to contact all 496 patients; 428 by telephone and 68 by mail. Our team made contact with 426 patients and found 53 patients had symptoms of a skin and soft tissue infection. Of these 53 patients, 36 (68.0%) agreed to participate in the present study. They were notified of a suspicious outbreak of M. abscessus infection following TPI and/or EB procedures at the clinic and the danger of the infection and interviewed by telephone with a standardized questionnaire.
- Case identification
- Thirty-six patients agreed to participate in the present study were classified into two categories. A confirmed case was defined as a SSTI diagnosed after 1 July 2008 and due to M. abscessus infection following a TPI and/or an EB at the clinic. A probable case was defined as a SSTI, diagnosed after 1 July 2008, following a TBI and/or an EB in the clinic that did not respond to standard antibacterial therapy.
- Active surveillance of patients by medical record survey and telephone who had visited the clinic since 1 July 2008 was conducted to define the extent of the outbreak. Board-certified physicians interviewed potential case patients using a standardized questionnaire to obtain data on demographic characteristics, pertinent details of the TPI and/or EB, additional procedures afterwards, the date of symptom onset, symptoms and treatment. The study protocol was approved by the Institutional Review Board at KCDC.
- Environmental investigation
- On 9 March 2009, 22 environmental specimens were collected from the clinic for mycobacterial culture. Specimens were taken from all devices and materials within the clinic that could have come into contact with the patient's skin before, during and after the TPI and/or EB procedure session, including pillows, beds, blankets, humidifier, air conditioner, front desk, telephone and refrigerator. Water samples were taken from the sink, water bucket, bathroom, and faucets. The samples, including disposable syringes, water for injection, drugs for injection may not accurately represent the environmental conditions at the time of infection.
- Laboratory testing
- Clinical specimens were obtained from 6 patients. In 2 patients, pus and exudates samples were collected from the infected wounds of the thigh and waist by sterile swabs respectively. In 2 patients, pus sample were collected from the infected wound of the waist and neck by needle-aspiration respectively. In the remaining 2, in addition, excision skin biopsies were done from the thigh and chest.
- Specimens were prepared as described previously [13,14]. Species identification for these isolates was done by means of comparative sequence analyses of portions of the 16S rRNA gene, hsp65 and rpoB, which were amplified by polymerase chain reaction (PCR) [15,16].
- Randomly amplified polymorphic DNA (RAPD) PCR, using three primers, INS-2, IS986-FP, URP-6, as described by Zhang et al. [17] with modification was performed on patient and environmental samples.
- Samples then underwent pulsed-field gel electrophoresis (PFGE) analysis. The large restriction fragments were divided and digested with Asel, as previously described [18].
- Isolates were tested for susceptibility against amikacin, cefoxitin, ciprofloxacin, clarithromycin, doxycycline, imipenem, linezolid, tobramycin and moxifloxacin by means of a broth microdilution method at the National Institute of Health [19].
- TPI and EB procedure description
- Twenty-one patients (58.3%) were disinfected with 75% ethanol swabs before a TPI and 17 patients (47.2%) were disinfected again after the TPI with 75% ethanol swabs. Before an EB, the skin was swabbed with povidone iodine and was not re-swabbed after an EB. After the skin was swabbed with 75% ethanol, a TPI was performed using disposable needles without time to dry. Physician applied the antiseptic using concentric circles from the center toward the outside. He used a glove only his left hand without performing hand hygiene. Epidural needle and spinal set were recycled after sterilization by autoclave. Drugs used for the TPIs were lidocaine, normal saline and dexamethasone. Lidocaine and normal saline were used for the EBs. High-frequency therapy was administered after a TPI, but not after an EB. Hot pack s were not used with any patients
MATERIALS AND METHODS
- Case patients
- The clinic was opened from 7 May 2005 to 1 March 2009. Most patients visited the clinic with neuromuscular pains. Of the 36 patients interviewed, Thirteen (36%) were male and 23 (64%) were female. The median age was 56 years (range 34 to 81 years). Underlying diseases were diabetes (n=4, 11.1%), hypertension (n=4, 11.1%), cerebral infarction (n=1, 2.8%) and malignant disease (n=1, 2.8%).
- Of the 36 patients, 29 (80.6%) underwent combined TPI and EB procedures at the clinic. Five (13.9%) underwent TPI only, while two (5.6%) underwent an unknown procedure. M. abscessus was isolated from the wound swab and needle aspiration of two patients respectively (5.6%). The average number of needle passages per day was as follows: 1 to 9 (27 patients, 75.0%) and 10 and over (9 patients, 25.0%). Five patients (13.9%) were treated with high-frequency therapy after their TPI and EB procedures.
- Thirty-six patients who underwent TPIs and/or EBs between 1 July 2008 and 28 February 2009 subsequently had skin lesions or abscesses at the injection sites. The outbreak ceased at the end of February 2009 due to the closure of the clinic (Figure 1).
- Clinical characteristics
- Ten patients (27.8%) had systemic symptoms, such as a fever or chills, and two patients (5.6%) had lymphadenopathy around their skin lesions. Out of 36 patients with papular skin lesions, 23 complained of a burning sensation around the skin lesion and 35 had erythematous papular skin lesions. Thirty-six patients (100%) had skin lesions corresponding to the injection site. The lesion sites were located as follows: thigh (n=20, 55.6%), waist (n=12, 33.3%), knee (n=12, 33.3%), shoulder (n=9, 25.0%), buttocks (n=7, 19.4%), hand (n=5, 13.9%), back (n=2, 5.6%), wrist (n=2, 5.6%) and neck (n=1, 2.8%). In addition, the mean number of lesions was 2.9 (range 0 to 30). The mean incubation time was 5.5 days (range 1 to 15 days). Clinical characteristics are summarized in Table 1.
- Environmental investigation
- The environmental samples were obtained on 9 March 2009. The samples may not have accurately represented the environmental conditions at the time of the outbreak because the clinic was closed due to private problems before the investigation. M. abscessus was not isolated from the 22 environmental samples taken from the clinic. However, most cultures were contaminated by various nosocomial pathogens, including several NTM species such as Mycobacterium septicum, M. avium and M. mageritense.
- Microbiologic examination
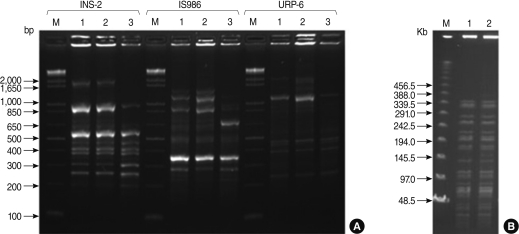
- Six specimens were obtained from the 6 patients respectively in four different hospitals (A, B, C and D). Mycobacteria were isolated from the wounds of two patients in two hospitals respectively (B and C). For these isolates, nucleotide sequences of the portions of the 16S rRNA, hsp65 and rpoB were determined and analyzed to identify the isolates as M. abscessus (Data not shown). Two isolates were identified as M. abscessus, with the same sequence analysis. Two isolates also were determined by RAPD-PCR and the isolates were identified as M. abscessus (Figure 2A).
- One was a patient's specimen which was collected from the wound swab in hospital B on 9 March 2009. The other specimen was collected from the wound needle aspiration in another hospital C. M. abscessus isolates from the two patients' skin wounds were compared using RAPD-PCR and PFGE analysis. RAPD-PCR and PFGE analysis resulted in an identical pattern among two M. abscessus strains (Figure 2B), implying that a single strain of M. abscessus was involved in the outbreak. The other four specimens were not identified a M. abscessus.
- Both of the isolates showed identical antimicrobial susceptibility profiles. They were sensitive to amikacin and clarithromycin, but resistant to doxycycline and ciprofloxacin.
- Treatment and outcomes
- Thirty-three (91.7%) of 36 patients were admitted to the different hospitals. Among them, 13 patients (39.4%) had undergone incision and drainage and two patients (6.1%) underwent aspiration of the lesions. The other 18 patients (54.5%) were given only antibiotic therapy. Ten of 18 patients were treated administration of oral clarithromycin (500 mg, twice daily) for 4-6 months and the other patients also received either amikacin or a combination of clarithromycin and ciprofloxacin. All admitted patients recovered fully during the investigation and no deaths occurred.
RESULTS
- Outbreaks of mycobacteria have been reported following injections of histamines, lidocaine, saline solution, vaccines, disinfectant solution and adrenal cortex extracts [10,20,21]. Injection safety is an enormous global health challenge both in the developing world, where safe injection practices are often lacking, and in developed countries, where new technologies and the transition of clinical practice to less regulated outpatient settings is occurring [22-24]. With the increase of TPI and/or EB procedures, adverse events also have been reported, such as needle pain, tiredness, bleeding, headache, faintness, and rarely, pneumothorax [25,26].
- Thirty-six patients who underwent TPIs and/or EBs at a same rural clinic between 1 July 2008 and 28 February 2009 subsequently had skin lesions or abscesses at the injection sites The symptoms seen in our cases is similar to that reported in previous outbreaks of M. abscessus infection following acupuncture in Korea [14,27]. Erythematous nodule or mass, a finding reportedly suggestive of M. abscessus infection following acupuncture [14], were seen in almost all of our patients. In our cases, M. abscessus was isolated from two patients only in two hospitals (B and C). Two strains of M. abscessus from patients were identical using PFGE analysis. Moreover, the 2 isolates were compared with use of RAPD-PCR with reference to type strain of M. abscessus (M. abscessus ATCC 19977) because almost 50% of strains cannot be assessed by PFGE [17]. Two methods resulted in an identical pattern among the 2 isolates.
- There were several limitations in identifying the exact contamination source. Because the clinic was closed due to private problems before the completion of the investigation, the environmental sources for the rapidly growing mycobacteria and their mechanism of distribution could not be elucidated.
- However, there are several possible sources of the outbreak. First, inadequate sterilization of the injection sites could be the source of the infection. Because 75% ethanol and povidone iodine have a wide-spectrum germicidal effect, they are commonly used as a disinfectant for procedures. When improperly used, however, the mixture is not as effective as a disinfectant. A considerable amount of time is necessary for the prevention of mycobacterial infections after 75% ethanol or povidone iodine is applied [28]. Second, insufficient equipment sterilization and the clinic's general environment could be the source of the infection. The equipment, including epidural needle, spinal set were sterilized with an autoclave by a nurse who works in a clinic without precise protocol of sterilization. A third possibility is improper preparation of the injection drugs and septic techniques. Drugs used for the TPIs were lidocaine, normal saline and dexamethasone, but dexamethasone was not used for the EBs. Since the middle of October 2008, the physician performed the TPIs drugs, including lidocaine and normal saline, except dexamethasone. After finishing the treatment of the last patient each day, the physician prepared the following day's injection drugs and placed them in a refrigerator for up to two days. When the outbreak began, the physician had been preparing the injection drugs and placing them in a refrigerator for up to two days. The drugs were not sterilized. The samples, including disposable syringes, water for injection, drugs for injection may not accurately represent the environmental conditions at the time of infection because the clinic was closed due to private problems before the completion of the investigation. Although contaminated drugs were not determined as the source of the outbreak, the long-term storage of the injection drugs could be implicated. Additionally, whenever the physician performed TPI and/or EB, he used a glove only his left hand without performing hand hygiene; creating yet another potential source of infection. Moreover, the physician responsible for the injections used multiple-dose bottles (100 mL) of normal saline in order to dilute lidocaine and dexamethasone. Yuan et al. [11] reported cases of post-injection abscesses from extrinsic contamination of multiple-dose bottles of normal saline.
- Another limitation was the difficulty in conducting the epidemiological study. Most of the 36 patients with symptoms had recovered before the study, so we obtained only six specimens in four hospitals (A, B, C and D). Thirty-three (91.7%) of 36 patients were treated in different hospitals, which limited our ability to obtain treatment methodology and results. We did not investigate whether the outbreak had started prior to 1 July 2008, because the physician did not report any patients with adverse effects before then. We could not compare patients with controls due to lack of consent and participation in this present study.
- In conclusion, we report an outbreak of M. abscessus infection following TPI and/or EB that involved 36 patients. There needs to be more attention for invasive procedures management, including TPI and EB in rural clinics. Epidemiologic studies also are needed to explain the exact source of infection to prevent future outbreaks of M. abscessus infection.
DISCUSSION
ACKNOWLEDGEMENTS
ACKNOWLEDGEMENTS
-
No author has a commercial or other association that might pose a conflict of interest.
-
This article is available from: http://e-epih.org/.
NOTES
- 1. Phillips MS, von Reyn CF. Nosocomial infections due to nontuberculous mycobacteria. Clin Infect Dis 2001;33:1363-1374. 11550115.ArticlePubMed
- 2. Primm TP, Lucero CA, Falkinham JO 3rd. Health impacts of environmental mycobacteria. Clin Microbiol Rev 2004;17:98-106. 14726457.ArticlePubMedPMC
- 3. Norton CD, LeChevallier MW. A pilot study of bacteriological population changes through potable water treatment and distribution. Appl Environ Microbiol 2000;66:268-276. 10618235.ArticlePubMedPMC
- 4. Falkinham JO 3rd. Nontuberculous mycobacteria in the environment. Clin Chest Med 2002;23:529-551. 12370991.ArticlePubMed
- 5. Moore M, Frerichs JB. An unusual acid-fast infection of the knee with subcutaneous, abscess-like lesions of the gluteal region: report of a case with a study of the organism, Mycobacterium abscessus, n. sp. J Invest Dermatol 1953;20:133-169. 13035193.ArticlePubMed
- 6. Fitzgerald DA, Smith AG, Lees A, Yee L, Cooper N, Harris SC, et al. Cutaneous infection with Mycobacterium abscessus. Br J Dermatol 1995;132:800-804. 7772489.ArticlePubMed
- 7. Ga H, Koh HJ, Choi JH, Kim CH. Intramuscular and nerve root stimulation vs lidocaine injection to trigger points in myofascial pain syndrome. J Rehabil Med 2007;39:374-378. 17549328.ArticlePubMed
- 8. Song CW, Kim JW. The effect of trigger point injection and C2-ganglion block for the patients with chronic headache. J Korean Pain Soc 1995;8:272-278 (Korean).
- 9. Wong CS, Wong SH. A new look at trigger point injections. Anesthesiol Res Pract 2012;2012:492452. 21969825.ArticlePubMedPMCPDF
- 10. Villanueva A, Calderon RV, Vargas BA, Ruiz F, Aguero S, Zhang Y, et al. Report on an outbreak of postinjection abscesses due to Mycobacterium abscessus, including management with surgery and clarithromycin therapy and comparison of strains by random amplified polymorphic DNA polymerase chain reaction. Clin Infect Dis 1997;24:1147-1153. 9195073.ArticlePubMed
- 11. Yuan J, Liu Y, Yang Z, Cai Y, Deng Z, Qin P, et al. Mycobacterium abscessus post-injection abscesses from extrinsic contamination of multiple-dose bottles of normal saline in a rural clinic. Int J Infect Dis 2009;13:537-542. 19269204.ArticlePubMed
- 12. Inman PM, Beck A, Brown AE, Stanford JL. Outbreak of injection abscesses due to Mycobacterium abscessus. Arch Dermatol 1969;100:141-147. 5797954.ArticlePubMed
- 13. Dytoc MT, Honish L, Shandro C, Ting PT, Chui L, Fiorillo L, et al. Clinical, microbiological, and epidemiological findings of an outbreak of Mycobacterium abscessus hand-and-foot disease. Diagn Microbiol Infect Dis 2005;53:39-45. 16054324.ArticlePubMed
- 14. Koh SJ, Song T, Kang YA, Choi JW, Chang KJ, Chu CS, et al. An outbreak of skin and soft tissue infection caused by Mycobacterium abscessus following acupuncture. Clin Microbiol Infect 2010;16:895-901. 19694761.ArticlePubMed
- 15. Adékambi T, Drancourt M. Dissection of phylogenetic relationships among 19 rapidly growing Mycobacterium species by 16S rRNA, hsp65, sodA, recA and rpoB gene sequencing. Int J Syst Evol Microbiol 2004;54:2095-2105. 15545441.ArticlePubMed
- 16. Kim HY, Kook Y, Yun YJ, Park CG, Lee NY, Shim TS, et al. Proportions of Mycobacterium massiliense and Mycobacterium bolletii strains among Korean Mycobacterium chelonae-Mycobacterium abscessus group isolates. J Clin Microbiol 2008;46:3384-3390. 18753344.ArticlePubMedPMC
- 17. Zhang Y, Rajagopalan M, Brown BA, Wallace RJ Jr. Randomly amplified polymorphic DNA PCR for comparison of Mycobacterium abscessus strains from nosocomial outbreaks. J Clin Microbiol 1997;35:3132-3139. 9399508.ArticlePubMedPMC
- 18. Jönsson BE, Gilljam M, Lindblad A, Ridell M, Wold AE, Welinder-Olsson C. Molecular epidemiology of Mycobacterium abscessus, with focus on cystic fibrosis. J Clin Microbiol 2007;45:1497-1504. 17376883.ArticlePubMedPMC
- 19. National Committee for Clinical Laboratory Standards. Susceptibility testing of mycobacteria, nocardiae, and other aerobic actinomycetes; approved standard. 2003. Wayne, PA: NCCLS.
- 20. Zhibang Y, BiXia Z, Qishan L, Lihao C, Xiangquan L, Huaping L. Large-scale outbreak of infection with Mycobacterium chelonae subsp. abscessus after penicillin injection. J Clin Microbiol 2002;40:2626-2628. 12089291.ArticlePubMedPMC
- 21. Galil K, Miller LA, Yakrus MA, Wallace RJ Jr, Mosley DG, England B, et al. Abscesses due to mycobacterium abscessus linked to injection of unapproved alternative medication. Emerg Infect Dis 1999;5:681-687. 10511524.ArticlePubMedPMC
- 22. Hutin YJ, Chen RT. Injection safety: a global challenge. Bull World Health Organ 1999;77:787-788. 10593025.PubMedPMC
- 23. Hutin YJ, Hauri AM, Armstrong GL. Use of injections in healthcare settings worldwide, 2000: literature review and regional estimates. BMJ 2003;327:1075. 14604927.ArticlePubMedPMC
- 24. Siegel JD, Rhinehart E, Jackson M, Chiarello L. Health Care Infection Control Practices Advisory Committee. 2007 Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Health Care Settings. Am J Infect Control 2007;35:S65-S164. 18068815.ArticlePubMedPMC
- 25. Shafer N. Pneumothorax following "trigger point" injection. JAMA 1970;213:1193. 5468270.Article
- 26. Elias M. Cervical epidural abscess following trigger point injection. J Pain Symptom Manage 1994;9:71-72. 8021535.ArticlePubMed
- 27. Song JY, Sohn JW, Jeong HW, Cheong HJ, Kim WJ, Kim MJ. An outbreak of post-acupuncture cutaneous infection due to Mycobacterium abscessus. BMC Infect Dis 2006;6:6. 16412228.ArticlePubMedPMC
- 28. Woo PC, Leung KW, Wong SS, Chong KT, Cheung EY, Yuen KY. Relatively alcohol-resistant mycobacteria are emerging pathogens in patients receiving acupuncture treatment. J Clin Microbiol 2002;40:1219-1224. 11923335.ArticlePubMedPMC
REFERENCES
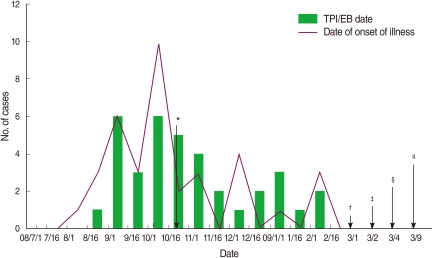
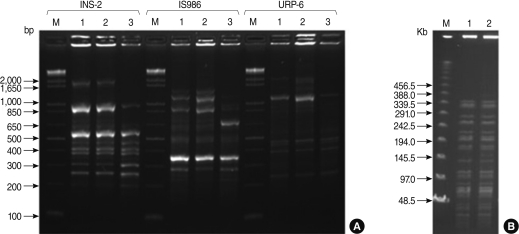
Figure & Data
References
Citations

- Nanomolar effective report of tetra-cationic silver(II) porphyrins against non-tuberculous mycobacteria in antimicrobial photodynamic approaches
Kevim Bordignon Guterres, Grazielle Guidolin Rossi, Marli Matiko Anraku de Campos, Kelly Schneider Moreira, Thiago Augusto Lima Burgo, Bernardo Almeida Iglesias
Photodiagnosis and Photodynamic Therapy.2022; 38: 102770. CrossRef - Purulent Flexor Tendon Rupture of the Hand due to Mycobacterium abscessus Infection: A Case Report and Review of the Literature
Yuji Tomori, Toru Mochizuki, Hiroko Ohno, Mitsuhiko Nanno, Tokifumi Majima
Journal of Nippon Medical School.2022; 89(3): 347. CrossRef - MYCOBACTERIOSIS: A REVIEW OF EVIDENCE-BASED CLINICAL MANIFESTATIONS AMONG HUMANS
F.S. PETROVA, I.V. PETROV, T.KH. AMIROVA, L.V. PETROVA
AVICENNA BULLETIN.2020; : 484. CrossRef - Severe hypercalcemia in a patient with extrapulmonaryMycobacterium abscessus: granuloma or immune reconstitution inflammatory syndrome? First case ofMycobacterium abscessuspresenting as retroperitoneal lymphadenopathy with severe hypercalcemia: a case rep
Omar Abdulfattah, Ebad Ur Rahman, FNU Shweta, Praveen Datar, Zainab Alnafoosi, David Trauber, Mirela Sam, Danilo Enriquez, Frances Schmidt
Journal of Community Hospital Internal Medicine Perspectives.2018; 8(6): 331. CrossRef - Mycobacteriological characteristics and treatment outcomes in extrapulmonary Mycobacterium abscessus complex infections
Suk Hyeon Jeong, Su-Young Kim, Hee Jae Huh, Chang-Seok Ki, Nam Yong Lee, Cheol-In Kang, Doo Ryeon Chung, Kyong Ran Peck, Sung Jae Shin, Won-Jung Koh
International Journal of Infectious Diseases.2017; 60: 49. CrossRef - Comparison of Saramis 4.12 and IVD 3.0 Vitek MS Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry for Identification of Mycobacteria from Solid and Liquid Culture Media
Caroline Leyer, Guillaume Gregorowicz, Faiza Mougari, Laurent Raskine, Emmanuelle Cambau, Dominique de Briel, Geoffrey A. Land
Journal of Clinical Microbiology.2017; 55(7): 2045. CrossRef - An outbreak of joint and cutaneous infections caused by non-tuberculous mycobacteria after corticosteroid injection
Sun-Young Jung, Bong Gi Kim, Donghyok Kwon, Ji-Hyuk Park, Seung-Ki Youn, Semi Jeon, Hye-Yeon Um, Kyoung-Eun Kwon, Hyun-Jung Kim, Hyun-Joo Jung, Eunmi Choi, Byung-Joo Park
International Journal of Infectious Diseases.2015; 36: 62. CrossRef - A Case of Meningitis Caused by Mycobacterium abscessus in a Paediatric Patient
D. R. Gayathri Devi, H. B. Mallikarjuna, Anusha Chaturvedi, S. Vishnu Prasad
Journal of Tuberculosis Research.2015; 03(02): 54. CrossRef - A Case of Continuous Ambulatory Peritoneal Dialysis-Associated Peritonitis Caused byMycobacterium abscessus
Hea Won Ann, Yong In Cho, Sang Hoon Han, Jun Yong Choi, Young Goo Song, Kyu Hun Choi, June Myung Kim
Korean Journal of Medicine.2014; 87(1): 110. CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite