Articles
- Page Path
- HOME > Epidemiol Health > Volume 42; 2020 > Article
-
Original Article
Factors associated with in-hospital death in patients with nosocomial infections: a registry-based study using community data in western Iran -
Salman Khazaei1
 , Erfan Ayubi2
, Erfan Ayubi2 , Ensiyeh Jenabi3
, Ensiyeh Jenabi3 , Saeid Bashirian4
, Saeid Bashirian4 , Masud Shojaeian5
, Masud Shojaeian5 , Leili Tapak6,7
, Leili Tapak6,7
-
Epidemiol Health 2020;42:e2020037.
DOI: https://doi.org/10.4178/epih.e2020037
Published online: June 1, 2020
1Research Center for Health Sciences, Hamadan University of Medical Sciences, Hamadan, Iran
2Department of Community Medicine, School of Medicine, Zahedan University of Medical Sciences, Zahedan, Iran
3Autism Spectrum Disorders Research Center, Hamadan University of Medical Sciences, Hamadan, Iran
4Social Determinants of Health Research Center, Hamadan University of Medical Sciences, Hamadan, Iran
5Deputy of Health, Hamadan University of Medical Sciences, Hamadan, Iran
6Department of Biostatistics, School of Public Health, Hamadan University of Medical Sciences, Hamadan, Iran
7Noncommunicable Diseases Research Center, Hamadan University of Medical Sciences, Hamadan, Iran
- Correspondence: Leili Tapak Department of Biostatistics, School of Public Health, Hamadan University of Medical Sciences, Shariati Crossroad, Hamadan 6517838678, Iran E-mail: l.tapak@umasha.ac.ir
©2020, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES
- Determining the predictors of in-hospital death related to nosocomial infections is an essential part of efforts made in the overall health system to improve the delivery of health care to patients. Therefore, this study investigated the predictors of in-hospital death related to nosocomial infections.
-
METHODS
- This registry-based, longitudinal study analyzed data on 8,895 hospital-acquired infections (HAIs) in Hamadan Province, Iran from March 2017 to December 2019. The medical records of all patients who had been admitted to the hospitals were extracted from the Iranian Nosocomial Infections Surveillance Software. The effects of the type and site of infection, as well as age group, on in-hospital death were estimated using univariate and multivariable Cox regression models.
-
RESULTS
- In total, 4,232 (47.8%) patients with HAIs were males, and their mean age was 48.25±26.22 years. In both sexes, most nosocomial infections involved Gram-negative bacteria and the most common site of infection was the urinary tract. Older patients had a higher risk of in-hospital death (adjusted hazard ratio [aHR], 2.26; 95% confidence interval [CI], 1.38 to 3.69 for males; aHR, 2.44; 95% CI, 1.29 to 4.62 for females). In both sexes, compared with urinary tract infections, an increased risk of in-hospital death was found for ventilator-associated events (VAEs) (by 95% for males and 93% for females) and bloodstream infections (BSIs) (by 67% for males and 82% for females).
-
CONCLUSIONS
- We found that VAEs, BSIs, and fungal infections were independently and strongly associated with increased mortality.
- Nosocomial infections (NIs) are among the leading causes of mortality and morbidity in hospitals, affecting hundreds of millions of patients around the world [1]. NIs increase hospital costs through the additional use of drugs—especially antibiotics—and by increasing the length of patients’ stay in the hospital [2].
- The World Health Organization defines NIs as infections occurring in a patient in a hospital or other healthcare facility, in whom the infection was not present nor incubating at the time of admission, as well as infections that are acquired during the stay but appear after discharge [3].
- Globally, a huge number of patients experience NIs, with incidence rates ranging from 3.5% to 12.0% in developed countries and from 5.7% to 19.1% in low-income and middle-income countries [4]. In a 1-day point prevalence study involving 1,265 intensive care units (ICUs) from 76 countries (the Extended Prevalence of Infection in Intensive Care study), 51% of patients were found to have NIs [5]. However, the variation reported in the literature can be attributed to the setting, the type of hospital, the patient population, and the precise definitions and surveillance techniques used in different societies [6]. In developing countries, the rates of nosocomial infections are over 8 times higher than in ICUs in the United States [7]. Evidence shows that the implementation of effective programs regarding NI surveillance can reduce the infection by approximately one-third [8].
- It has been reported that bacteremia, lung disease, and multisite infection, as well as pneumonia in intensive care patients, are associated with an increased risk of mortality in patients with NIs [9,10]. The results of a multicenter study in France showed that 39%, 20%, and 14% of all NIs were attributed to lower respiratory tract, bloodstream, and surgical wound infections, respectively [11]. According another study in Norway, bloodstream infections (BSIs), lower respiratory tract infections, and multiple simultaneous infections were predictors of death in patients with NIs [12].
- Carrying out studies to determine the predictors of mortality and to analyze serious untoward events during the provision of health services is an essential part of the efforts made by the overall health system to improve the delivery of health care to patients. Therefore, the aim of this study was to investigate the predictors of in-hospital NI-associated death.
INTRODUCTION
- This registry-based, longitudinal study was conducted using data on 8,895 hospital-acquired infections (HAIs) in Hamadan Province in western Iran from March 2017 to December 2019. Hamadan Province encompasses an area of 19,493 km2 and had a population of 1,758,268 people according to the national census held in 2016 by Statistical Center of Iran [13].
- We used information on HAIs from the following hospitals: Alimoradian in Nahavand; Besat, Shahid-Beheshti, Sina, and Fatemieh in Hamadan; Vali-Asr in Tuyserkan; Ghaem in Asadabad; Imam Hossein and Mehr in Malayer; Imam Reza in Kabudarahang; Valiar in Razan; and Imam Reza in Kabudarahang, all of which recorded information on their infection rates in the Iranian Nosocomial Infections Surveillance Software (INISS).
- Patients with clinical signs of an NI 48 hours after admission with no sign of bacterial colonization at the time of admission were enrolled in the study. The medical records of all patients who had been admitted to the hospital in the abovementioned time period were extracted from the INISS. We abstracted data from the software and recorded information including the demographic characteristics of patients (e.g., age and sex), year of occurrence, type of infection (Gram-negative bacterial, Gram-positive bacterial, viral, or fungal), site of infection (bone and joint; cardiovascular system; central nervous system; eye, ear, nose, or throat; gastrointestinal system; reproductive tract; and lower respiratory tract), and the ward of admission. Patients’ information was entered into the INISS daily, based on the findings of a physical examination and the patient’s signs and symptoms. The completeness and accuracy of the medical records was assessed by the infection control staff of the corresponding hospitals.
- Statistical analysis
- The descriptive results are expressed as frequencies (%) or as mean±standard deviation. The survival of patients with NIs according to the type and site of infection was investigated using Kaplan-Meier survival analysis. The log-rank test was applied to evaluate the statistical significance of differences in survival curves. The effects of the type and site of infection, as well as age groups, on in-hospital death were estimated using univariate and multivariable Cox regression models and the results presented as hazard ratios (HRs) and 95% confidence intervals (CIs). The proportional hazards (PH) assumption of the variables included in the model was assessed using Schoenfeld residuals. A p-value < 0.05 was considered to indicate statistical significance. All statistical analyses were performed using Stata version 14 (StataCorp., College Station, TX, USA).
- Ethics statement
- This study was approved by Ethics Committee of Hamadan University of Medical Sciences (IR.UMSHA.REC.1399.012).
MATERIALS AND METHODS
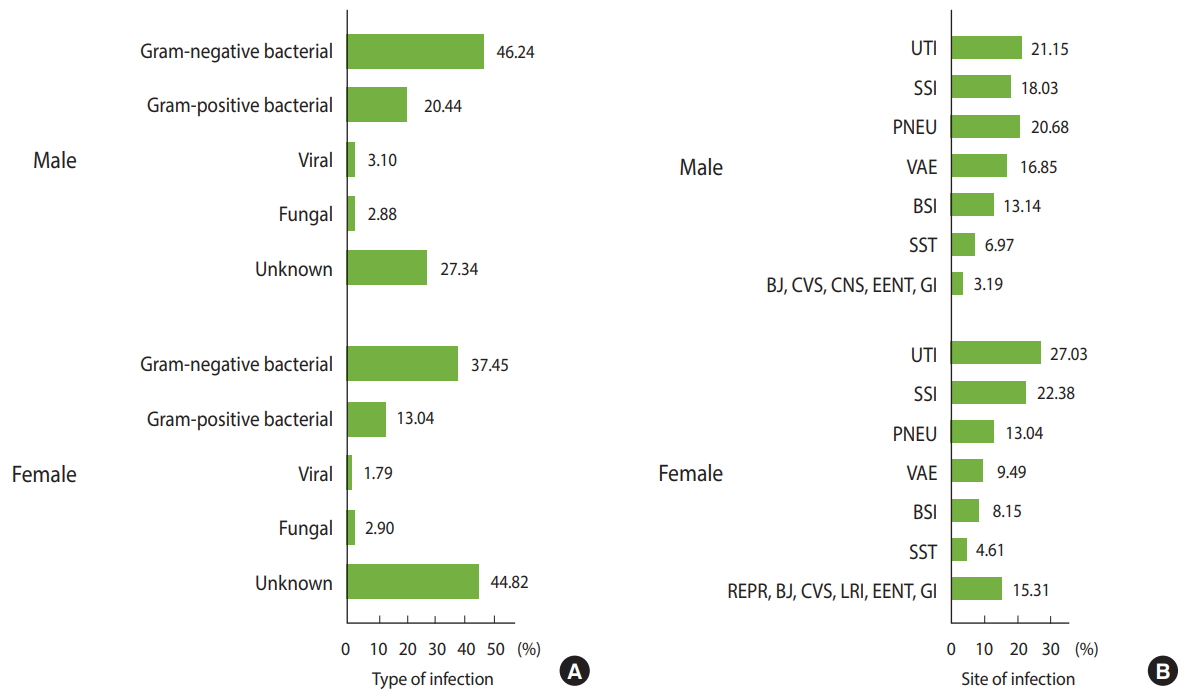
- In total, 8,895 patients with NIs were identified, of whom 4,232 (47.8%) were males (Table 1). Their mean age was 48.25±26.22 years (range, < 1 to 99). The mean length of hospital stay was 21.26±38.95 days (range, < 1 to 439) and the corresponding figures for time of admission to HAI and from HAI to discharge or death were 12.46±27.94 days (range, < 1 to 390) and 10.97±24.39 days (range, < 1 to 375), respectively. In both sexes, most NIs resulted from Gram-negative bacteria, followed by the Gram-positive bacteria and viral and fungal infections. By site, the most common infections were urinary tract infections (UTIs), followed by surgical site infections (SSIs), pneumonia (PNEU), ventilator-associated events (VAE), BSIs, skin and soft tissue infections, and infections of other organs (Figure 1). The distribution of NIs by type and site in both sexes according to age group are presented in Supplementary Materials 1 and 2.
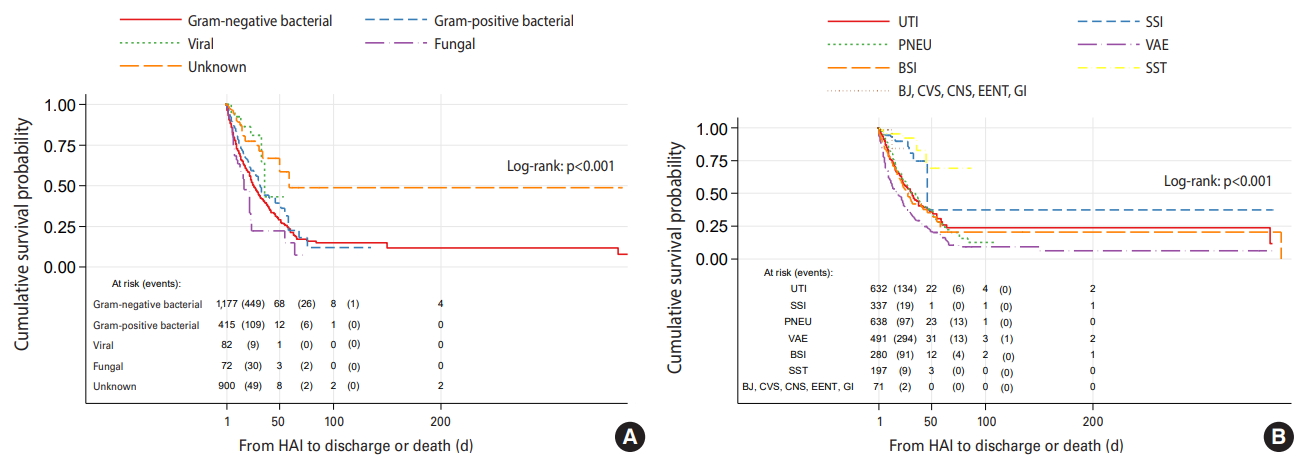
- The effects of the type and site of HAIs on survival, as well as the results of sex-specific univariate Cox regression analyses, are shown in Table 2, Figures 2 and 3. Between March 2017 and December 2019, among 1,283 (20.5%) in-hospital deaths, 717 (55.8%) were in male patients and 566 (44.2%) were in female patients. Compared to Gram-negative bacterial infection, the crude HR of fungal infections for death was 1.40 (95% CI, 0.98 to 2.01; p=0.06) (Table 2). Applying Kaplan-Meier curves, survival was poorer in male cases with nosocomial fungal infections than in those with other types of infections (log-rank test, p<0.001) (Figure 2A), as the median survival for fungal infections was 17days (95% CI, 11 to 23), whereas the corresponding figure for Gram-negative bacterial infections was 26 days (95% CI, 23 to 30) (Figure 2A). Among male patients, the HR (95% CI) of VAEs for mortality compared to UTIs was 1.80 (95% CI, 1.47 to 2.19; p<0.001) (Table 2). Male with VAEs had shorter survival than those with other infections (log-rank test, p<0.001) with a median survival of 18 days (95% CI, 14 to 22) (Figure 2B).
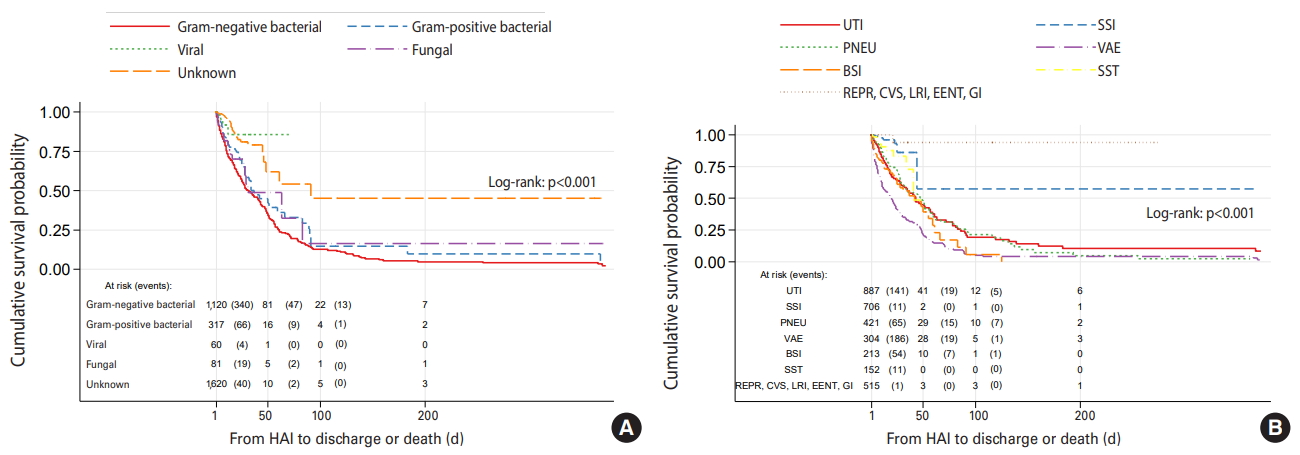
- Female patients with fungal and Gram-negative bacterial NIs had significantly worse survival than those with other types of infections, with a median survival of 31 days (95% CI, 27 to 83) and 32 days (95% CI, 27 to 40), respectively (log-rank test, p<0.001) (Figure 3A). Similarly to male patients, female patients with VAEs had significantly shorter survival than those with NIs at other sites (log-rank test, p<0.001) with a median survival of 20 days (95% CI, 14 to 24) (Figure 2B). Compared to UTIs, the crude HR for mortality associated with VAEs was 1.93 (95% CI, 1.56 to 2.39; p<0.001) (Table 2). The Schoenfeld residuals test did not suggest violation of the PH assumption for the variables included in the analysis.
- The results of the multivariable Cox regression analysis are presented in Table 2. Older patients (> 65 years) had a higher risk of in-hospital death than younger patients (15-24 years), with aHRs of 2.26 (95% CI, 1.38 to 3.69) for males and 2.44 (95% CI, 1.29 to 4.62) for females. Among male patients with fungal NIs, an 87% increase was found in the risk of in-hospital death when compared to those with Gram-negative bacterial infections. Among both sexes, compared with UTIs, an increased risk of in-hospital death was found for VAEs (by 95% for males and 93% for females) and BSIs (by 67% for males and 82% for females). Although, SSIs were associated with a lower risk of in-hospital death—by 50% for males and 69% for females—these associations were not statistically significant.
RESULTS
- This study was conducted to identify the predictors of in-hospital death related to NIs. The main findings in this study were that in both sexes, bacteria were the most common cause of NIs. Moreover, by site, UTIs were most common, followed by SSIs and PNEU. Older patients had a higher risk of in-hospital death. Fungal NIs were associated with a higher risk of in-hospital death than Gram-negative bacterial infections in males. Among both sexes, compared with UTIs, VAEs, and BSIs were associated with a higher risk of in-hospital death, while SSIs were associated with a lower risk of in-hospital death.
- Bacteria are responsible for roughly 90% of NIs, whereas other microorganisms make less of a contribution to the development of NIs [2]. This may be because some bacteria belong to the natural flora of the patient and cause an infection only when the immune system of the patient is weakened. We likewise found that bacteria were the leading cause of NIs in hospitalized patients in Hamadan Province. The results of the present study indicate that the most common NIs in inpatients of both sexes were UTIs. In 2007, an analysis of the INISS showed that UTIs were the most common NI in Iran, accounting for 32.2% of cases, but in that year, UTIs were most common in females, whereas PNEU was most common in males [14]. Differences in patterns of using invasive procedures for therapeutic and diagnostic purposes, implementing chemotherapy programs for cancer, and performing organ transplants in hospitals may cause differences in the distribution of NIs across sites and hospitals [15].
- We found that in both sexes, most NIs resulted from Gramnegative bacteria. Gram-negative bacteria are bacteria that do not retain the crystal violet stain used in the Gram staining method of bacterial differentiation. Gram-negative bacilli cause the 4 most frequent types of HAI, including PNEU, SSIs, UTIs, and BSIs, which are caused by Klebsiella, Acinetobacter, Pseudomonas aeruginosa, and Escherichia coli, as well as many other less common bacteria [16]. Consistent with our findings, in the United States Gram-negative bacteria are responsible for more than 30% of HAIs, whereas in the ICUs of the United States, Gram-negative bacteria account for about 70% of these types of infections [17]. These bacteria are often resistant to multiple drugs and are increasingly resistant to most available antibiotics [18].
- In accordance with our findings, Koch et al. [12] showed that old age was associated with an increased risk of death in patients with NIs. Defective host defenses and the prevalence of many chronic diseases such as cancer, diabetes, and atherosclerosis are partly responsible for the increased frequency of infections and death in older patients [19]. Differences in immunity status and other comorbidities can be associated with differences in the distribution of sites of infection according to age and, consequently, in the risk of death. Avci et al. [20] showed that the most common site of infection in elderly patients was the urinary tract, whereas in non-elderly patients it was the lower respiratory tract. The incidence of UTIs, respiratory tract infections, SSIs, skin and soft tissue infections, primary bacteremia, and prosthesis infections was significantly higher in elderly patients.
- In accordance with our results, other studies have also shown that patients with BSIs had an increased risk of death [21,22]. We also found that the mortality risk for patients with SSIs was lower than others, a result that has also been observed in other studies [22,23].
- The present study had some limitations. First, the sensitivity of the NI surveillance system in Iran is relatively low and does not cover all patients [14]. A second limitation of this study is the lack of access to denominator data to calculate the incidence of NIs by age group, sex, and microorganism type. Finally, it was not possible in this study to investigate illness severity as a possible predictor of death.
- We found that VAEs, BSIs, and fungal infections were independently and strongly associated with increased mortality in hospitalized patients.
DISCUSSION
Electronic Supplementary Material
-
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare for this study.
-
FUNDING
Hamadan University of Medical Sciences funded this study.
-
AUTHOR CONTRIBUTIONS
Conceptualization: SK, LT. Data curation: SK, EA, EJ. Funding acquisition: MS. Methodology: SK, LT, EA, EJ, MS, SB. Project administration: SK, LT. Visualization: SK, LT. Writing – original draft: SK, LT, EA, EJ, MS, SB. Writing – review & editing: SK, LT, EA, EJ, MS, SB.
NOTES
ACKNOWLEDGEMENTS



| Characteristics |
Male (n=4,232) |
Female (n=4,627) |
||
|---|---|---|---|---|
| No (n=2,041) | Yes (n=717) | No (n=2,943) | Yes (n=566) | |
| Age (yr) | ||||
| 15-24 | 113 (5.5) | 17 (2.4) | 369 (12.5) | 10 (1.8) |
| 0-14 | 397 (19.4) | 16 (2.2) | 304 (10.3) | 5 (0.9) |
| 25-54 | 582 (28.5) | 153 (21.3) | 1,433 (48.7) | 70 (12.4) |
| 55-64 | 303 (14.8) | 104 (14.5) | 273 (9.3) | 80 (14.1) |
| ≥ 65 | 646 (31.6) | 427 (59.5) | 564 (19.2) | 401 (70.8) |
| Type of infection | ||||
| Gram-negative bacterial | 725 (35.5) | 505 (70.4) | 740 (25.1) | 414 (73.1) |
| Gram-positive bacterial | 308 (15.1) | 119 (16.6) | 250 (8.5) | 80 (14.1) |
| Viral | 79 (3.9) | 9 (1.3) | 59 (2.0) | 4 (0.7) |
| Fungal | 41 (2.0) | 32 (4.5) | 61 (2.1) | 24 (4.2) |
| Unknown | 888 (43.5) | 52 (7.5) | 1,833 (62.3) | 44 (7.8) |
| Site of infection | ||||
| UTI | 510 (25.0) | 146 (20.4) | 750 (25.5) | 172 (30.4) |
| SSI | 341 (16.7) | 19 (2.6) | 790 (26.8) | 11 (1.9) |
| PNEU | 534 (26.2) | 113 (15.8) | 339 (11.5) | 91 (16.1) |
| VAE | 186 (9.1) | 320 (44.6) | 96 (3.3) | 213 (37.6) |
| BSI | 190 (9.3) | 108 (15.1) | 156 (5.3) | 67 (11.8) |
| SST | 204 (10.0) | 9 (1.3) | 163 (5.5) | 11 (1.9) |
| Other1 | 76 (3.7) | 2 (0.3) | 649 (22.0) | 1 (0.2) |
Values are presented as number (%).
UTI, urinary tract infection; SSI, surgical site infection; PNEU, pneumonia; VAE, ventilator-associated events; BSI, bloodstream infection; SST, skin and soft tissue infection
1 Bone and joint, cardiovascular system, central nervous system, eye, ear, nose, throat, or mouth, and gastrointestinal infections for males; Reproductive tract, bone and joint, cardiovascular system, lower respiratory infections, and gastrointestinal infections for females.
| Variables |
Male |
Female |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| cHR (95% CI) | p-value | aHR (95% CI) | p-value | cHR (95% CI) | p-value | aHR (95% CI) | p-value | ||
| Age (yr) | |||||||||
| 15-24 | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | |||||
| 0-14 | 0.51 (0.26, 1.02) | 0.060 | 0.54 (0.27, 1.08) | 0.080 | 0.45 (0.15, 1.32) | 0.150 | 0.27 (0.09, 0.79) | 0.020 | |
| 25-54 | 1.45 (0.88, 2.41) | 0.140 | 1.40 (0.84, 2.32) | 0.180 | 1.25 (0.64, 2.44) | 0.500 | 1.02 (0.52, 1.99) | 0.940 | |
| 55-64 | 1.81 (1.08, 3.04) | 0.020 | 1.49 (0.89, 2.51) | 0.130 | 3.86 (1.99, 7.46) | <0.001 | 2.06 (1.06, 4.01) | 0.030 | |
| ≥ 65 | 2.79 (1.72, 4.53) | <0.001 | 2.26 (1.38, 3.69) | 0.001 | 5.25 (2.79, 9.86) | <0.001 | 2.44 (1.29, 4.62) | 0.006 | |
| Type of infection | |||||||||
| Gram-negative bacterial | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | |||||
| Gram-positive bacterial | 0.78 (0.63, 0.95) | 0.020 | 0.93 (0.75, 1.15) | 0.520 | 0.72 (0.56, 0.91) | 0.008 | 0.87 (0.67, 1.12) | 0.290 | |
| Viral | 0.32 (0.16, 0.61) | 0.001 | 0.87 (0.43, 1.76) | 0.710 | 0.29 (0.11, 0.77) | 0.010 | 0.81 (0.28, 2.27) | 0.690 | |
| Fungal | 1.40 (0.98, 2.01) | 0.060 | 1.87 (1.28, 2.74) | 0.001 | 0.75 (0.48, 1.17) | 0.210 | 1.11 (0.71, 1.75) | 0.630 | |
| Unknown | 0.31 (0.23, 0.41) | <0.001 | 0.58 (0.42, 0.81) | 0.001 | 0.17 (0.13, 0.24) | <0.001 | 0.59 (0.40, 0.86) | 0.007 | |
| Site of infection | |||||||||
| UTI | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | |||||
| SSI | 0.31 (0.19, 0.50) | <0.001 | 0.50 (0.30, 0.83) | 0.008 | 0.15 (0.08, 0.28) | <0.001 | 0.31 (0.16, 0.60) | 0.001 | |
| PNEU | 0.86 (0.67, 1.10) | 0.230 | 1.14 (0.87, 1.49) | 0.330 | 0.96 (0.74, 1.24) | 0.750 | 1.03 (0.78, 1.36) | 0.810 | |
| VAE | 1.80 (1.47, 2.19) | <0.001 | 1.94 (1.57, 2.40) | <0.001 | 2.15 (1.75, 2.64) | <0.001 | 1.93 (1.56, 2.39) | <0.001 | |
| BSI | 1.17 (0.90, 1.52) | 0.220 | 1.67 (1.26, 2.22) | <0.001 | 1.40 (1.04, 1.87) | 0.020 | 1.82 (1.34, 2.46) | <0.001 | |
| SST | 0.20 (0.10, 0.40) | <0.001 | 0.40 (0.20, 0.83) | 0.010 | 0.46 (0.25, 0.85) | 0.010 | 0.87 (0.45, 1.71) | 0.700 | |
| Other1 | 0.25 (0.06, 1.01) | 0.050 | 0.48 (0.11, 1.99) | 0.310 | 0.02 (0.00, 0.15) | <0.001 | 0.06 (0.00, 0.45) | 0.006 | |
cHR, crude hazard ratio; aHR, adjusted hazard ratio; UTI, urinary tract infection; SSI, surgical site infection; PNEU, pneumonia; VAE, ventilator-associated events; BSI, bloodstream infection; SST, skin and soft tissue infection
1 Bone and joint, cardiovascular system, central nervous system, eye, ear, nose, throat, or mouth, and gastrointestinal infections for males; Reproductive tract, bone and joint, cardiovascular system, lower respiratory infections, and gastrointestinal infections for females.
- 1. Morillo-García Á, Aldana-Espinal JM, Olry de Labry-Lima A, Valencia-Martín R, López-Márquez R, Loscertales-Abril M, et al. Hospital costs associated with nosocomial infections in a pediatric intensive care unit. Gac Sanit 2015;29:282-287.ArticlePubMed
- 2. Khan HA, Ahmad A, Mehboob R. Nosocomial infections and their control strategies. Asian Pac J Trop Biomed 2015;5:509-514.Article
- 3. World Health Organization. Prevention of hospital-acquired infections: a practical guide; 2002 [cited 2020 Jun 9]. Available from: https://www.who.int/csr/resources/publications/whocdscsreph200212.pdf.
- 4. Bagheri Nejad S, Allegranzi B, Syed SB, Ellis B, Pittet D. Healthcare-associated infection in Africa: a systematic review. Bull World Health Organ 2011;89:757-765.ArticlePubMedPMC
- 5. Vincent JL, Rello Condomines J, Marshall J, Silva E, Anzueto A, Martin CD, et al. The extended prevalence of infection in the ICU study: EPIC II. JAMA 2009;302:1-49.Article
- 6. Gastmeier P, Sohr D, Just HM, Nassauer A, Daschner F, Rüden H. How to survey nosocomial infections. Infect Control Hosp Epidemiol 2000;21:366-370.ArticlePubMed
- 7. Rosenthal VD, Al-Abdely HM, El-Kholy AA, AlKhawaja SA, Leblebicioglu H, Mehta Y, et al. International Nosocomial Infection Control Consortium report, data summary of 50 countries for 2010-2015: device-associated module. Am J Infect Control 2016;44:1495-1504.ArticlePubMed
- 8. Haley RW, Culver DH, White JW, Morgan WM, Emori TG, Munn VP, et al. The efficacy of infection surveillance and control programs in preventing nosocomial infections in US hospitals. Am J Epidemiol 1985;121:182-205.ArticlePubMedPDF
- 9. Moine P, Timsit JF, De Lassence A, Troché G, Fosse JP, Alberti C, et al. Mortality associated with late-onset pneumonia in the intensive care unit: results of a multi-center cohort study. Intensive Care Med 2002;28:154-163.ArticlePubMedPDF
- 10. Decoster A, Grandbastien B, Demory MF, Leclercq V, Alfandari S; Regional Network ‘Review of nosocomial infection-related mortality’. A prospective study of nosocomial-infection-related mortality assessed through mortality reviews in 14 hospitals in Northern France. J Hosp Infect 2012;80:310-315.ArticlePubMed
- 11. Kaoutar B, Joly C, L’Hériteau F, Barbut F, Robert J, Denis M, et al. Nosocomial infections and hospital mortality: a multicentre epidemiology study. J Hosp Infect 2004;58:268-275.ArticlePubMed
- 12. Koch AM, Nilsen RM, Eriksen HM, Cox RJ, Harthug S. Mortality related to hospital-associated infections in a tertiary hospital; repeated cross-sectional studies between 2004-2011. Antimicrob Resist Infect Control 2015;4:57.ArticlePubMedPMCPDF
- 13. Statistical Center of Iran. Iran data portal: census 2016. [cited 2020 Mar 1]. Available from: http://irandataportal.syr.edu/census/census-2016.
- 14. Zahraei SM, Eshrati B, Masoumi Asl H, Pezeshki Z. Epidemiology of four main nosocomial infections in Iran during March 2007 - March 2008 based on the findings of a routine surveillance system. Arch Iran Med 2012;15:764-766.PubMed
- 15. Bereket W, Hemalatha K, Getenet B, Wondwossen T, Solomon A, Zeynudin A, et al. Update on bacterial nosocomial infections. Eur Rev Med Pharmacol Sci 2012;16:1039-1044.PubMed
- 16. Weinstein RA. Epidemiology and control of nosocomial infections in adult intensive care units. Am J Med 1991;91:179S-184S.ArticlePubMed
- 17. Gaynes R, Edwards JR; National Nosocomial Infections Surveillance System. Overview of nosocomial infections caused by gram-negative bacilli. Clin Infect Dis 2005;41:848-854.ArticlePubMedPDF
- 18. Centers for Disease Control and Prevention. Gram-negative bacteria infections in healthcare settings; 2011 [cited 2020 Mar 1]. Available from: https://www.cdc.gov/hai/organisms/gram-negative-bacteria.html.
- 19. Maia Fde O, Duarte YA, Lebrão ML, Santos JL. Risk factors for mortality among elderly people. Rev Saude Publica 2006;40:1049-1056 (Portuguese).ArticlePubMedPDF
- 20. Avci M, Ozgenc O, Coskuner SA, Olut AI. Hospital acquired infections (HAI) in the elderly: comparison with the younger patients. Arch Gerontol Geriatr 2012;54:247-250.ArticlePubMed
- 21. Vrijens F, Hulstaert F, Devriese S, van de Sande S. Hospital-acquired infections in Belgian acute-care hospitals: an estimation of their global impact on mortality, length of stay and healthcare costs. Epidemiol Infect 2012;140:126-136.ArticlePubMed
- 22. Fabbro-Peray P, Sotto A, Defez C, Cazaban M, Molinari L, Pinède M, et al. Mortality attributable to nosocomial infection: a cohort of patients with and without nosocomial infection in a French university hospital. Infect Control Hosp Epidemiol 2007;28:265-272.ArticlePubMed
- 23. Kanerva M, Ollgren J, Virtanen MJ, Lyytikäinen O; Prevalence Survey Study Group. Risk factors for death in a cohort of patients with and without healthcare-associated infections in Finnish acute care hospitals. J Hosp Infect 2008;70:353-360.ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Predictors of In-ICU Mortality Among Older Patients with Healthcare-Associated Infection: A Cohort Study
Fereshteh Rezaie, Farahnaz Mohammadi-Shahboulaghi, Reza Fadayevatan, Mohsen Shati, Gholamreza Ghaedamini Harouni
Journal of Kermanshah University of Medical Sciences.2023;[Epub] CrossRef - Barriers to hand hygiene compliance in intensive care units to prevent the spread of healthcare-associated infections
Gurjeet Singh, Raksha Singh, Ranga Reddy Burri
MGM Journal of Medical Sciences.2023; 10(4): 667. CrossRef - Prevalence of Nosocomial Infections During the COVID-19 Pandemic: A Systematic Review and Meta-analysis
Sima Rafiei, Zahra Nejatifar, Rana Soheylirad, Samira Raoofi, Fatemeh Pashazadeh Kan, Ahmad Ghashghaee
Journal of Health Reports and Technology.2022;[Epub] CrossRef - Prevalence of Nosocomial Infections During the COVID-19 Pandemic: A Systematic Review and Meta-analysis
Sima Rafiei, Zahra Nejatifar, Rana Soheylirad, Samira Raoofi, Fatemeh Pashazadeh Kan, Ahmad Ghashghaee
Journal of Health Reports and Technology.2022;[Epub] CrossRef - The Impact of COVID-19 Outbreak on Nosocomial Infection Rate: A Case of Iran
Maryam Jabarpour, Mahlagha Dehghan, Giti Afsharipour, Elham Hajipour Abaee, Parvin Mangolian Shahrbabaki, Mehdi Ahmadinejad, Mahboobeh Maazallahi, Aseer Manilal
Canadian Journal of Infectious Diseases and Medical Microbiology.2021; 2021: 1. CrossRef - Survival rate in patients with ICU-acquired infections and its related factors in Iran’s hospitals
MEDSKorosh Etemad, Yousef Khani, Seyed-Saeed Hashemi-Nazari, Neda Izadi, Babak Eshrati, Yadollah Mehrabi
BMC Public Health.2021;[Epub] CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite